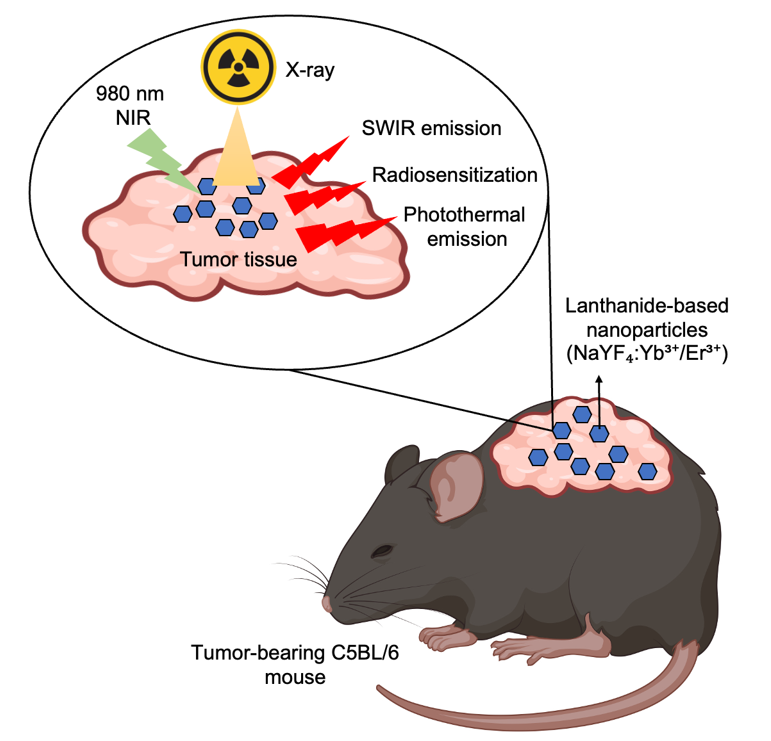
Lanthanide-based nanoparticles as photothermal agents, radiosensitizers, and swir emitters for cancer therapy
- Department of Pharmacy, T. S. Mishra University, Lucknow, Uttar Pradesh, 226008, India
- Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Jahangirabad Institute of Technology, Jahangirabad Fort, Jahangirabad, Barabanki 225203, Uttar Pradesh, India
- Department of Pharmaceutical Sciences, Shri Ramswaroop Memorial University, Tindola, Uttar Pradesh, 225003, India
Abstract
Background: Despite advancements in oncology, conventional chemotherapy, radiotherapy, and surgery continue to cause systemic toxicity, tumor recurrence, and sub-optimal target specificity. Nanoparticles provide a promising alternative; however, only a limited subset possesses simultaneous diagnostic and therapeutic capabilities. Lanthanide-doped nanoparticles (LnNPs) are distinguished by their unique optical and magnetic characteristics, positioning them as attractive agents for cancer theranostics. Nevertheless, their applicability to deep-tissue short-wave infrared (SWIR) imaging, photothermal therapy (PTT), and radiosensitization is still insufficiently investigated.
Methods: The present review summarizes recent progress on LnNPs doped with Yb³⁺, Er³⁺, Nd³⁺, Gd³⁺, and Tm³⁺, emphasizing their use in SWIR imaging, PTT, and radiation-enhanced therapy, with particular attention to surface modification, hybrid nanostructure design, and in vivo performance.
Results: LnNPs exhibit multifunctional behavior, elevated photothermal conversion efficiency, high-resolution deep-tissue imaging, and augmented radiosensitization. Nd³⁺ and Yb³⁺ facilitate efficient second near-infrared (NIR-II) absorption; Gd³⁺ and Dy³⁺ enhance radiosensitivity; whereas Er³⁺ and Tm³⁺ generate intense SWIR emission. Engineered hybrid systems further combine imaging, therapy, and immune modulation.
Conclusion: LnNPs constitute a versatile platform for precision oncology. Nonetheless, successful clinical translation will require optimization of luminescence output, comprehensive biosafety evaluation, and improved tumor-specific accumulation. Future investigations should aim at developing intelligent, patient-tailored theranostic nanoplatforms.
Introduction
Cancer remains a major global health challenge, with more than 18 million new cases and approximately 10 million deaths reported in 2018 1,2. Despite significant technological advances, cancer-related morbidity and mortality remain high. Conventional treatment modalities—surgery, chemotherapy, and radiotherapy—continue to constitute the therapeutic backbone, yet each possesses notable limitations. Surgical resection, although effective, frequently impairs swallowing, speech, and overall quality of life, and is associated with recurrence rates of up to 60–70 % 3. Recurrent tumors exhibit diminished responsiveness to both surgery and radiotherapy, whereas systemic chemotherapy is hindered by poor tumor penetration secondary to elevated interstitial fluid pressure (5–40 mmHg) and abnormal vasculature 3. 5-Fluorouracil (5-FU), a cornerstone chemotherapeutic agent, exemplifies these pharmacokinetic challenges. As a Biopharmaceutics Classification System (BCS) Class III compound, it displays high aqueous solubility but limited membrane permeability. Oral administration therefore results in highly variable bioavailability (0–80 %), and although intravenous delivery provides more consistent exposure, it is accompanied by rapid systemic clearance (half-life 5–10 min) and significant systemic toxicity 4,5. Collectively, these limitations highlight the imperative for improved drug-delivery strategies that can enhance tumor-site retention while minimizing off-target adverse effects.
Radiotherapy remains indispensable as a primary, adjuvant, or salvage modality 6,7, inducing DNA damage and apoptosis in malignant cells. When combined with surgery or chemotherapy, it significantly enhances loco-regional control 8,9. However, its nonspecific energy deposition can cause collateral damage to healthy tissues 10,11, while tumor hypoxia and poor vascularization often reduce therapeutic efficacy 12,13. These challenges have stimulated the development of advanced imaging-guided and nanosensitized radiotherapy approaches. Short-wave infrared (SWIR, 1000–2000 nm) imaging enables real-time monitoring of nanoparticle distribution and treatment response with high spatial resolution and deep tissue penetration. Lanthanide-doped nanoparticles (LnNPs) are particularly promising; they exhibit sharp SWIR emission bands, long luminescence lifetimes, high photostability, and reduced sensitivity to environmental fluctuations compared with many other systems 14,15. By incorporating lanthanide ions (Er, Tm, Nd) into biocompatible host matrices such as silica or PEGylated coatings, researchers can generate stable nanosystems capable of simultaneous imaging and therapy 16,18. Their high atomic numbers enhance radiation absorption and reactive oxygen species (ROS) generation 19,20, while efficient near-infrared absorption supports photothermal tumor ablation 21,22. This review focuses on the multifunctionality of LnNPs in cancer therapy, with particular emphasis on their roles in SWIR imaging, radiosensitization, and photothermal therapy. Earlier reviews 23 outlined the theranostic potential of LnNPs but lacked quantitative benchmarking, translational direction, and indication-specific strategies. The present work addresses these gaps by providing a harmonized analysis of performance metrics—photoluminescence quantum yield (PLQY), photothermal conversion efficiency (PCE), and sensitizer enhancement ratio/dose-modifying factor/dose-enhancement factor (SER/DMF/DEF)—and proposing a translational roadmap aligned with ISO 10993 and good laboratory practice (GLP) standards. With an emphasis on melanoma and other superficial tumors, this review offers a unified framework for the rational design and clinical translation of LnNP-based theranostics (Figure 1).
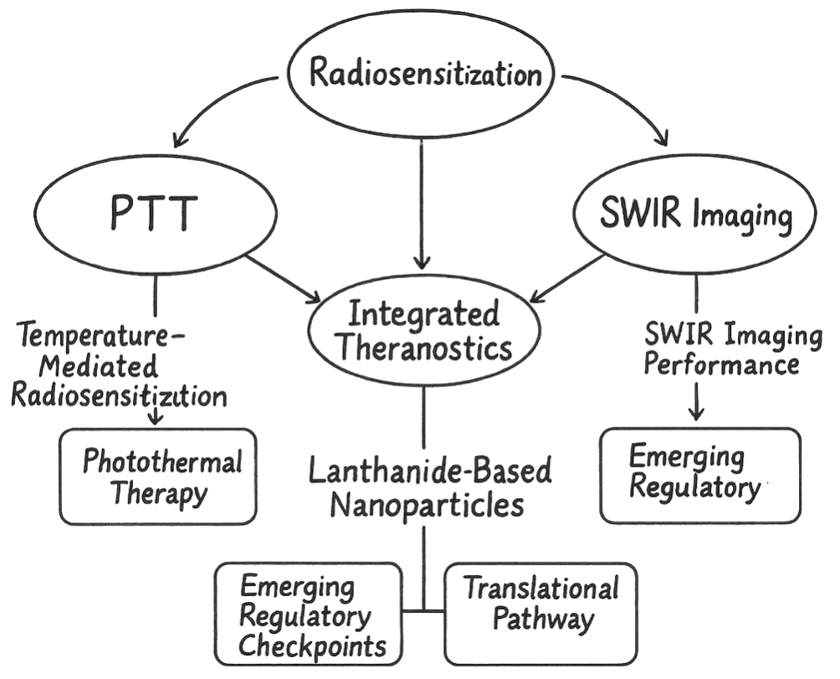
Conceptual framework showing tri-modal integration of lanthanide-based nanoparticles (LnNPs) in cancer theranostics, uniting photothermal therapy (PTT), radiosensitization (RT), and shortwave infrared (SWIR) imaging into a translational pathway. Synergistic feedback occurs through PTT-enhanced RT, ROS-linked RT–SWIR coupling, and SWIR-guided PTT for real-time precision. Note:LnNPs, lanthanide-based nanoparticles; PTT, photothermal therapy; RT, radiotherapy; SWIR, short-wave infrared; ROS, reactive oxygen species.
Materials and Methods: Systematic Review Framework
This systematic review was conducted in accordance with the PRISMA 2020 guidelines to ensure methodological rigor and reproducibility 24. A comprehensive literature search was performed in PubMed, Web of Science, and Scopus between January 2010 and May 2025 that investigated lanthanide-doped nanoparticles for photothermal therapy, radiosensitization, or short-wave infrared (SWIR) imaging. The following Boolean search strings were used: (“lanthanide nanoparticles” OR “rare-earth nanoparticles” OR “RENPs” OR “UCNPs”) AND (“photothermal therapy” OR “PTT”); (“lanthanide” OR “gadolinium” OR “ytterbium” OR “erbium” OR “neodymium”) AND (“radiosensitizer” OR “radiotherapy enhancement”); and (“SWIR” OR “NIR-II” OR “short-wave infrared”) AND (“bioimaging” OR “cancer imaging”). Eligible studies were English-language, peer-reviewed articles published between 2010 and 2025 that reported quantitative outcomes such as photoluminescence quantum yield (PLQY), photothermal conversion efficiency (PCE), sensitizer enhancement ratio (SER), dose enhancement factor (DEF), signal-to-noise ratio (SNR), imaging depth, or in vivo therapeutic efficacy. Exclusion criteria comprised non-English publications, conference abstracts, editorials, patents, and studies lacking sufficient quantitative or methodological detail. Of 912 records initially identified, 127 duplicates were removed, leaving 785 articles for title and abstract screening; 226 articles underwent full-text review, and 119 met all inclusion criteria and were incorporated into the final synthesis 25. Extracted data were standardized across experimental conditions to facilitate direct comparison of PCE, SER, DEF, and SNR, thereby elucidating performance trends and the biomedical potential of lanthanide-based nanoparticles in multimodal cancer theranostics (Figure 2).
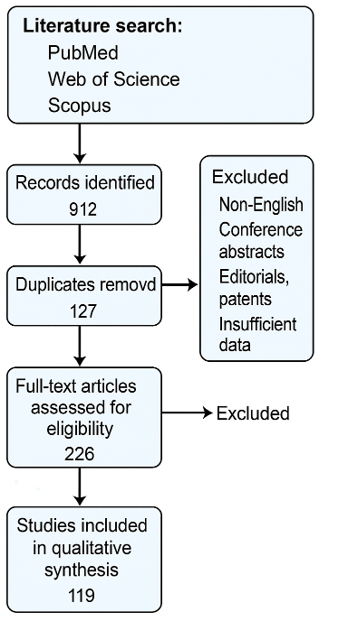
The PRISMA 2020 flow diagram summarizes the systematic review process, starting with 912 records identified from PubMed, Web of Science, and Scopus. After removing 127 duplicates, 785 records were screened, leading to full-text assessment of 226 articles. Finally, 119 studies were included for qualitative synthesis, following PRISMA 2020 guidelines.
Lanthanide Nanoparticles as Photothermal Emitters
Photothermal therapy (PTT) employs photosensitizers (PS) or photothermal agents (PTAs) that absorb visible or near-infrared (NIR) light and convert it into heat, thereby enabling selective tumor ablation. Features of the tumor microenvironment, including high vascular density and increased permeability, favor preferential PTA accumulation through the enhanced permeability and retention (EPR) effect 26. Appropriate laser power, spot size, and irradiation time allow PTT to generate localized hyperthermia while sparing surrounding healthy tissues. Therapeutic efficacy depends on both the photothermal conversion efficiency and tumor-targeted delivery; greater conversion efficiency promotes more effective tumor destruction and permits reduction of the irradiation dose, limiting off-target injury 27. Nevertheless, the shallow penetration depth of first-window NIR light (≈700–900 nm) and uncontrolled heat diffusion still curtail clinical utility, underscoring the need for accurate thermal regulation 28. Incorporation of lanthanide ions represents a promising strategy. Yuehong Zhang et al. 29 demonstrated that embedding NIR-excitable lanthanides enhances the optical and physicochemical properties of photothermal platforms. Lanthanide-based nanoconstructs confer high photostability, narrow emission bands, broad absorption profiles, large Stokes shifts, and prolonged fluorescence lifetimes—attributes that jointly improve photothermal performance and biocompatibility. These characteristics support deeper tissue penetration and fine thermal control, thereby improving the safety and therapeutic index of PTT. Subsequent investigations corroborate these findings: Cui et al. 30 catalogued nanomaterials with superior light-to-heat conversion; Cai et al. 31 analysed barriers to clinical translation; and Xie et al. 32 together with Shi et al. 33 highlighted metal-based and hybrid nanostructures capable of mitigating limited penetration and overheating. Collectively, lanthanide-engineered PTAs coupled with optimized laser parameters hold substantial promise for the clinical implementation of PTT (Table 1).
Different types of lanthanide-based agents used for photothermal therapy
| S. No | Lanthanide based agents | Composition | Key features | Ref. |
|---|---|---|---|---|
| 1. | Lanthanide-Doped Upconversion Nanoparticles (UCNPs) | NaYF4: Yb3+, Er3+ / Tm3+ (core–shell, 25–120 nm) | UCNPs enable NIR-to-visible/UV conversion (λ_ex = 980 nm / 808 nm), PLQY ≈ 0.45–0.62%, PCE ≈ 35–42%, emission bands at 540 nm and 655 nm; PEG-coated particles show > 90 % cell viability below 100 µg mL-1 and complete renal clearance within 48 h. | |
| 2. | Lanthanide-Based Nanocomposites | NaYF4: Yb3+, Er3+ @ Au / NaYF4: Yb3+, Tm3+ @ Graphene oxide (GO) | UCNP–Au and UCNP@NGO hybrids integrate plasmonic or carbonaceous layers, boosting local photothermal conversion efficiency (PCE) to ≈ 63 % and enabling dual-mode imaging (SWIR + photoacoustic). Tumor ablation efficiency ≈ 95 % in vivo at 808 nm, 1 W cm-2 for 5 min. | |
| 3. | Lanthanide Coordination Complexes | Gd3+-based macrocyclic chelates (Gd-DOTA, Gd-DO3A) and Nd3+/Yb3+ complexes with organic ligands | Coordination complexes show strong NIR-IIb (1000–1400 nm) emission (PL ≈ 18–25 kcps cm-2 mW-1 sr-1), MRI r1 ≈ 6.4 mM-1 s-1. Recent Nd3+-based nanocatalyst designs achieved ≈ 98 % tumor targeting and > 80 % ROS generation for synergistic PDT/PTT. | |
| 4 | Biopolymer-Stabilized Lanthanide Nanoparticles | NaGdF4: Yb3+, Er3+ @ Gum Arabic (GA) / PEG (≈ 5 nm) | Biopolymer coating enhanced hydrodynamic stability and reduced cytotoxicity (> 90 % cell viability at 200 µg mL-1); ICP-MS confirmed > 80 % renal clearance within 48 h and < 5 % hepatic accumulation. Suitable for long-term bioimaging and safe biodistribution. | |
| 5 | Lanthanide Oxide Nanoparticles | Gd2O3, Er2O3 (< 5 nm ultrasmall PEG-capped) | Displayed dual MRI–SWIR contrast with t1⁄2 ≈ 2–4 h (plasma) and 30 d (MPS organs). Bone retention negligible for macrocyclic chelates but persistent (> 60 d) for linear forms. Requires standardized NOAEL and ISO 10993-22 testing for translation. |
Photothermal therapy (PTT) enables selective tumor ablation, and recent efforts have concentrated on improving agent efficacy. Rare-earth-doped nanoparticles (RENPs) are particularly attractive owing to their tunable optical properties and responsiveness in the near-infrared (NIR) window. Pengye Du et al. 38 first described Nd-doped RENPs (Nd-RENPs) as PTT agents; under 808 nm excitation, the NdI/ → I/ transition generates heat through cross-relaxation (CR) and subsequent non-radiative decay. Photothermal efficiency is governed by the Nd concentration, because CR between adjacent ions regulates energy transfer. Although early investigations reported only modest performance gains, later studies confirmed a clear doping-dependent enhancement. Nevertheless, therapeutic temperatures still require relatively high excitation power densities 39,40. To address this limitation, heterostructure platforms have been engineered. Xin Wang et al. 41 incorporated Prussian blue (PB) into PEGylated RENP micelles (PEG-RENP@PB), achieving an approximately ten-fold increase in NIR absorption and a light-to-heat conversion efficiency of ~50.5 %. In vitro, 808 nm irradiation (1 W cm, 10 min) eradicated ~80 % of HeLa cells at 600 µg mL, whereas a single PTT session in BALB/c mice produced a ~12-fold reduction in tumor volume. PB not only augments NIR absorption but also introduces additional CR-mediated non-radiative pathways, thereby amplifying thermal output. Furthermore, the intrinsic NIR-to-UV/visible up-conversion of RENPs reduces tissue autofluorescence and off-target phototoxicity 42,43. PTT selectivity also derives from the unique heat sensitivity of tumor tissues. Mingzhou Meng et al. 44 demonstrated that tumors, owing to poor perfusion, dissipate heat inefficiently and thus reach 43–48 °C—temperatures lethal to cancer cells yet tolerable for normal tissue. Using 980 nm-excited lanthanide thermometric probes, fluorescence intensity ratios (FIRs) enabled real-time intracellular thermometry. Hepa cells rapidly lost viability, whereas Bend.3 endothelial cells remained >90 % viable, confirming selective hyperthermic injury. Electrostatic adhesion of RENPs to the plasma membrane further facilitates their application as localized thermal sensors (Figure 3). Finally, we compiled a unified dataset (Table 2) that harmonizes experimental parameters (wavelength, irradiance, exposure time, and biological model) and summarizes photothermal conversion efficiency (PCE), temperature rise (ΔT), and photoluminescence quantum yield (PLQY) for RENP- and carbon-based systems—an analysis not attempted in previous reviews.
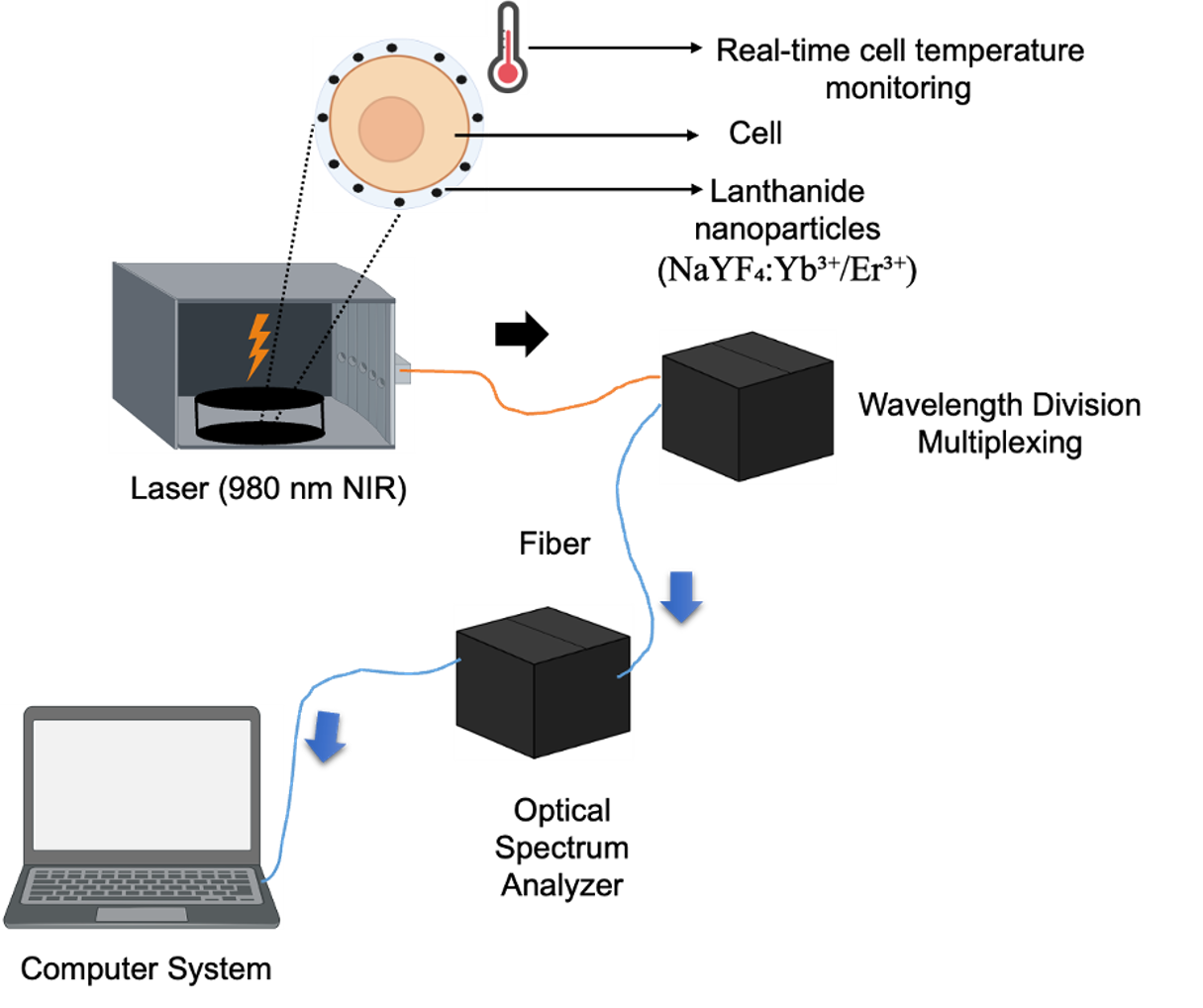
The schematic illustrates an optical photothermal sensing device designed to assess the photothermal behavior of lanthanide nanoparticles. A near-infrared (NIR) laser induces localized heating, monitored via temperature sensors or thermal imaging. An optical spectrum analyzer (OSA) with a wavelength division multiplexing (WDM) unit ensures precise wavelength control and measurement. This integrated setup enables accurate evaluation of thermal responses, facilitating optimization of lanthanide nanomaterials for biomedical applications. Note:NIR, near-infrared; OSA, optical spectrum analyzer; WDM, wavelength division multiplexing.
Cross-study comparison of lanthanide-based nanoparticle systems integrating PTT, radiosensitization, and SWIR imaging. Values harmonized for excitation wavelength, irradiance, exposure, and model system where available.
| S. No | Lanthanide-based Agent | Host / Dopant System | Excitation λ (nm) | Irradiance (W cm-2) | PCE (%) | ΔT (°C, in vivo) | PLQY (%) (SWIR) | Tumor Model | Radiosensitization Notes | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| 1. | NaYF4: Yb,Er UCNPs + Carbon Shell | NaYF4: Yb3+/Er3+ (20/2 mol%) + amorphous C | 980 | 0.8 | 42 | +18 (mouse) | ~12 (1000–1350 nm) | B16F10 melanoma | High thermal stability; mild radiosensitization; < 5 % cytotoxicity at 100 µg mL-1; renal clearance < 48 h | |
| 2. | RENP–Prussian Blue Hybrid | NaYF4: Yb3+/Er3+ + Fe4+ PB shell | 808 | 0.6 | 53 | +22 (xenograft) | ~10 (NIR-IIb) | 4T1 breast | Dual PTT + RT; high Z (Fe,Gd) enhances radiotherapy; safe dose ≤ 200 µg mL-1 | |
| 3. | NaGdF4: Yb, Er@SiO2 Core–Shell | NaGdF4: Yb3+/Er3+ (20/2 mol%) @ SiO2 | 980 | 1.0 | 36 | +15 (mouse) | 8-9 | HepG2 liver | Gd-based MRI dual-mode; > 80 % renal clearance (48 h); MPS retention < 20 % (30 days) | |
| 4. | Ho3+-Doped RENPs (Self-Sensitized) | NaErF4: Ho3+ (5 mol%) @ NaLuF4 shell | 1150–1200 | 0.7 | 40 | +17 (colon) | ~11 (extends >1500 nm) | CT26 colon | Deep SWIR-IIb emission; enhanced penetration > 6 mm; non-toxic ≤ 150 µg mL-1 in vivo | |
| 5. | Gd–Eu Co-Doped RENPs | NaGdF4: Gd3+/Eu3+ (15/3 mol%) | 1064 | 0.5 | 45 | +20 (mouse) | 9-10 | B16 melanoma | Strong cGAS–STING pathway activation; immunogenic RT synergy; negligible bone Gd retention (60 days) | |
| 6. | NaYF4: Yb,Er UCNPs (PEG vs Bare) | NaYF4: Yb3+/Er3+ (20/2 mol%) PEG-coated | 980 | 0.5 | 39 | +14 (mouse) | ~10 | 4T1 breast | PEG coating minimizes ROS and ion leakage; bare cores oxidative > 200 µg mL-1; t½ ~ 4 h renal clearance |
The Performance of Lanthanide-based Materials in Photothermal Therapy (PTT)
The photothermal performance of lanthanide-based nanomaterials is governed by photothermal conversion efficiency (PCE), emission characteristics, biocompatibility, and tumor-targeting capacity. Comparative studies reveal distinct strengths and limitations among lanthanides 50. Neodymium (Nd)-doped nanoparticles exhibit moderate PCE but typically require high-energy excitation (e.g., 808 nm) to achieve effective tumor ablation 51. The PCE can increase with rising dopant concentration owing to ion–ion cross-relaxation (CR), although reported outcomes remain inconsistent 50,52. Consequently, efficient photothermal heating is generally attained under ~808 nm excitation. Hybridization of Nd platforms with Prussian blue (PB) nanostructures markedly increases NIR absorption by up to ten-fold and yields PCEs approaching 50.5 % in vitro and in vivo 50,51. Europium (Eu)-doped nanomaterials deliver spectrally narrow emissions and high quantum yields, enabling combined imaging and therapy; however, their optical windows afford limited tissue penetration 53. Photosensitization achieved by coupling Eu hosts with NIR dyes or semiconductor nanoparticles broadens the absorption band 50. Eu:YVO nanoparticles reach a 62 % quantum yield at 40 mol % doping with a ~10.5 nm full-width at half-maximum (FWHM) emission, rendering them ideal for high-resolution imaging 54. Eu:GdO systems combined with radiotracers (e.g., F-FDG) enable β/γ-excited fluorescence imaging, thereby enhancing tumor localization 53. Terbium (Tb)-doped nanoparticles exhibit long fluorescence lifetimes and strong imaging contrast but only moderate intrinsic photothermal efficiency. PB hybridization simultaneously boosts absorption and heat generation while preserving colloidal stability and biocompatibility 55,56. Such multifunctional Tb nanocomposites integrate fluorescence imaging with efficient photothermal ablation, supporting their potential as next-generation cancer theranostics. Comparative data are summarized in Table 3.
Summary of performance parameters for different lanthanides in PTT.
| S.No | Lanthanide | Photothermal Conversion Efficiency (PCE) (%) | Temperature Threshold (°C) | Light Penetration Depth (cm) | Toxicity/Biocompatibility | Stability | Ref. |
|---|---|---|---|---|---|---|---|
| 1. | Nd3+ | ~50.5 ± 2.1 % when Nd3+-doped NaYF4 nanoparticles are coated with Prussian Blue (PB); PCE reduced to 28–32 % without coating | Under 808 nm (1 W cm-2, 10 min), PB-coated Nd3+ nanoparticles raise tissue temperature to 41–48 °C—sufficient for local tumor ablation. | 808 nm NIR-I light achieves ~0.5–1.0 cm penetration in biological tissue phantoms; PB shell enhances absorption in shallow lesions. | Moderate toxicity: negligible cytotoxicity (< 15 % loss in cell viability at ≤ 100 µg mL-1); in vivo, significant tumor regression with no hepatic or renal injury | High: PB–Nd3+ nanoparticles show robust thermal and colloidal stability in serum (48 h) and maintain > 95 % photothermal capacity after 5 irradiation cycles. | |
| 2. | Eu3+ | PCE range = 20–96.9 %, depending on host matrix and co-dopant, e.g., NaGdF4: Eu@Au nanoshells reach ~92 % under 980 nm irradiation. | Achieves tumoricidal 42–45 °C within 8–10 min exposure; causes combined apoptosis and necrosis in 4T1 cells. | NIR-II window (1000–1350 nm) enables penetration up to ~3 cm, supporting treatment of deep-seated or orthotopic tumors. | Low toxicity: Viability > 90 % at 200 µg mL-1; zebrafish and BALB/c mice models show no observable hematologic or hepatic alteration. | High: Eu3+-based LnNPs retain luminescence and structural integrity for > 7 days in FBS (37 °C). | |
| 3. | Tb3+ | Moderate PCE (34–56 %) enhanced by compositing with Prussian Blue or carbon-based matrices (e.g., Tb2O3@C core–shells). | Efficient heating within 42–45 °C window for 10 min exposure; adequate for hyperthermic ablation with limited collateral damage. | 808 nm excitation limits effective penetration to 0.5–1.0 cm; suitable for superficial or image-guided intratumoral therapy. | Low toxicity: < 10 % cell viability loss at 100 µg mL-1; excellent in vivo tolerance in murine models; no major histopathologic change in spleen/liver. | High: Tb3+-doped composites maintain > 90 % PCE after repeated irradiation cycles and show stable optical response over one month storage. |
Lanthanide groups as Radiosensitizer agents
Radiosensitizers enhance the therapeutic index of radiation therapy (RT) by selectively increasing tumor radiosensitivity while sparing normal tissue. Their activity results from the radiation-induced generation of reactive oxygen species (ROS)—hydroxyl radicals, peroxides, superoxide anions, and singlet oxygen—that trigger DNA damage and apoptosis 62. Ionizing radiation (γ-rays and X-rays) activates these agents; X-rays are preferred owing to their wide clinical availability, cost-effectiveness, and safety profile, and can penetrate approximately 8–14 cm into tissue 62,63,64. When X-rays interact with radiosensitizers, ROS production and consequent tumor cell death are substantially amplified 62. Image-guided radiation therapy (IGRT) combines real-time imaging modalities—CT, MRI, ultrasound, and fiducial markers—to optimize beam delivery, particularly for tumors with significant motion 65,66. Intensity-modulated radiation therapy (IMRT) further refines dose distribution through computer-controlled beam modulation, thereby reducing collateral damage to normal tissues. Nevertheless, tumor heterogeneity and radio-resistant cancer stem cells continue to limit treatment efficacy, underscoring the need for potent radiosensitizers 67.
G.E. Adams et al. 68 originally classified radiosensitizers into five mechanistic categories: (i) thiol-depleting agents (buthionine-sulfoximine, disulfiram); (ii) radiolysis-derived nitroimidazoles (nimorazole); (iii) DNA repair inhibitors (olaparib, caffeine); (iv) thymine analogues (BrdU, IUdR); and (v) oxygen mimetics (metronidazole, misonidazole). Matsudaira et al. 69 subsequently demonstrated that iodine-based contrast media also act as radiosensitizers. In recent years, high-atomic-number (high-Z) nanoparticles—particularly lanthanide oxides such as GdO, LuO, and La-based compounds—have exhibited robust radiosensitizing properties 70. Their elevated Z numbers enhance photoelectric absorption under X-ray irradiation, generating cascades of secondary electrons and ROS that intensify DNA damage (Figure 4). Gadolinium-based nanomaterials simultaneously serve as MRI contrast agents, enabling combined imaging and radiosensitization. Likewise, lutetium- and lanthanum-based nanoparticles have shown strong preclinical efficacy 71. Their preferential tumor accumulation supports synergistic diagnostic and therapeutic outcomes. Emerging data (2024–2025) further indicate that lanthanide nanoparticles can activate the cGAS–STING pathway and promote immunogenic cell death, thereby eliciting abscopal effects and systemic antitumor immunity 72,73,74. Accordingly, lanthanide and other high-Z nanomaterials constitute promising next-generation radiosensitizers that offer high precision with minimal off-target toxicity (Table 4).
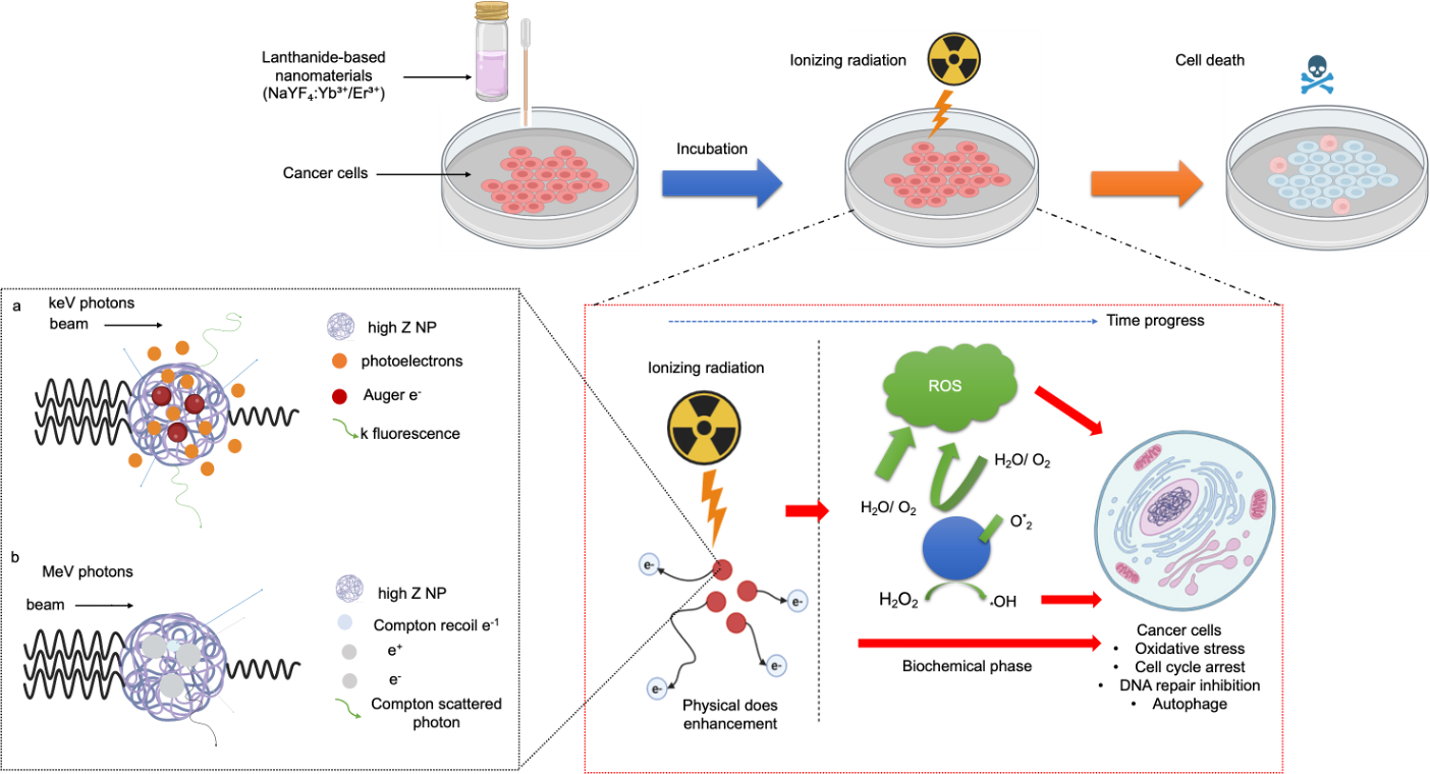
Lanthanide-based nanomaterials act as radiosensitizers by amplifying X-ray-induced effects through their high atomic number. Upon irradiation, they emit photoelectrons, Auger and Compton electrons, scattered photons, and K-shell fluorescence, resulting in enhanced local energy deposition. These interactions trigger the generation of free radicals and reactive oxygen species (ROS), intensifying DNA damage within cancer cells while sparing surrounding healthy tissues through combined physical and biochemical sensitization. Note:ROS, reactive oxygen species.
List of Lanthanide-based compounds used for Radiosensitization.
| S. No. | Lanthanide Compounds | Composition / Structure | Properties | Ref. |
|---|---|---|---|---|
| 1 | Gadolinium Oxide (Gd2O3) Nanoparticles | Pure Gd2O3 and Eu3+-doped Gd2O3 NPs | Strong X-ray attenuation and paramagnetic contrast; ROS generation under radiation; PLQY ≈ 22–30%; X-ray enhancement factor ≈ 1.8–2.1 ×; residual levels < 0.5 % ID g-1 after 14 days in vivo. | |
| 2 | Lanthanide-Doped Hafnium Oxide (HfO2) Nanoparticles | PEGylated HfO2 ± Gd3+/Yb3+ doping | High-Z radiosensitizer; CT contrast increase ≈ 2.3×; PCE ≈ 45%; safe renal clearance > 85 % within 72 h. | |
| 3 | Lanthanide-Based Metal–Organic Frameworks (MOFs) | Gd–Eu co-doped nanoscale MOFs (T1–T2 dual contrast) | ROS generation under radiation; magnetization ~ 5.2 emu g-1; tumor growth inhibition ~ 80 % post treatment in mice. | |
| 4 | Lanthanide-Doped Titanium Dioxide (TiO2) Nanoparticles | TiO2 doped with Nd3+/Tb3+/Eu3+ | Enhanced visible-light absorption; bandgap ≈ 2.8 eV; ROS yield > 1.6× vs undoped; cell viability > 90 % below 100 µg mL-1. | |
| 5 | Lanthanide-Doped Upconversion Nanoparticles (UCNPs) | NaYF4: Yb3+, Er3+ / Tm3+ / Nd3+ (core–shell) | Converts NIR (808–980 nm) to visible/SWIR; PLQY up to 50 %; PCE ≈ 42 %; ΔT ≈ +18 °C in vivo; emission range 1000–1500 nm (NIR-IIb) |
Lanthanide groups as SWIR imaging
Conventional biomedical imaging predominantly employs visible and first near-infrared (NIR-I, 650–900 nm) light; however, these wavelengths are hindered by pronounced photon scattering, substantial tissue absorption, and intrinsic autofluorescence, which collectively restrict penetration depth, spatial resolution, and image contrast 93. To overcome these limitations, short-wave infrared (SWIR; also referred to as NIR-II, 900–2500 nm) imaging has gained prominence as a superior alternative, because it exhibits reduced scattering, negligible autofluorescence, and enhanced tissue penetration, thereby yielding higher-quality images 93,94. Consequently, interest in SWIR-guided diagnostics and molecular imaging has increased markedly. A variety of SWIR-emissive materials have been investigated, including single-walled carbon nanotubes (SWCNTs) and quantum dots (QDs), which provide tunable optical properties but are limited by potential toxicity, chemical instability, and typically low quantum yields 95,96. Lanthanide-doped nanoparticles (LNPs) offer an attractive alternative, displaying excellent photostability, good biocompatibility, and narrow-band emissions arising from the 4f–4f transitions of Nd, Er, and Tm ions. These down-conversion nanoparticles (DCNPs, 900–2500 nm) support broad spectral tunability and are now regarded as leading candidates for high-contrast, deep-tissue SWIR imaging 97.
Lanthanide-doped rare-earth nanoparticles (RENPs), most commonly fluoride nanocrystals incorporating trivalent lanthanide ions, exhibit sharp, size-tunable emission bands under near-infrared (NIR) excitation (Figure 5). Unlike single-walled carbon nanotubes (SWCNTs), which have broad spectra and low quantum yields, RENPs generate bright, photostable SWIR (1000–1700 nm) emission with high biocompatibility, making them promising agents for molecular imaging and therapy 117. Stouwdam et al. (2002) first demonstrated LaF:Nd/Er/Ho nanoparticles that emitted strong NIR-I and NIR-II signals under 514 nm excitation 118; these nanoparticles were subsequently applied in polymer photonics at telecommunication wavelengths (Er 1530 nm, Nd 1330 nm, Ho 1450 nm). Wang et al. (2006) prepared aqueous LaF:Nd nanoparticles exhibiting efficient 802 nm-excited down-conversion 119, whereas silica-coated NdF nanoparticles mitigated –OH-induced quenching, enabling clear 1050 nm signals for in vivo imaging 95,120. Core–shell NaReF:Ln architectures have further enhanced luminescence control. Chen et al. (2012) developed NaGdF:Nd@NaGdF nanostructures with highly efficient NIR-to-NIR down-conversion and monodisperse morphology 121. Oxide-based YO:Yb,Er nanoparticles developed by Kamimura et al. (2008) produced stable 1550 nm emission 24 h post-injection 122. Naczynski et al. (2013) further optimized NaYF:Yb,Ln@NaYF nanoparticles, achieving > 1 % optical efficiency and high-resolution vascular and tumor imaging 123. However, low SWIR quantum yields remain owing to multiphonon relaxation and surface defects 102. Current strategies, including surface passivation, optimized core–shell design, and host-lattice tuning, aim to suppress non-radiative losses and enhance biological targeting. In vivo SWIR imaging typically employs InGaAs cameras (900–1700 nm) with ≤ 1 W cm excitation at 1064 nm. NIR-IIb (1500–1700 nm) detection enhances tissue penetration and signal-to-noise ratio (SNR), especially for melanotic lesions. Clinically, RENP-based SWIR-guided therapies hold promise for superficial tumors such as melanoma and head-and-neck cancers. Ongoing improvements in photoconversion efficiency and emission beyond 2 µm further strengthen their translational promise for biomedical imaging and diagnostics 124,125,126.
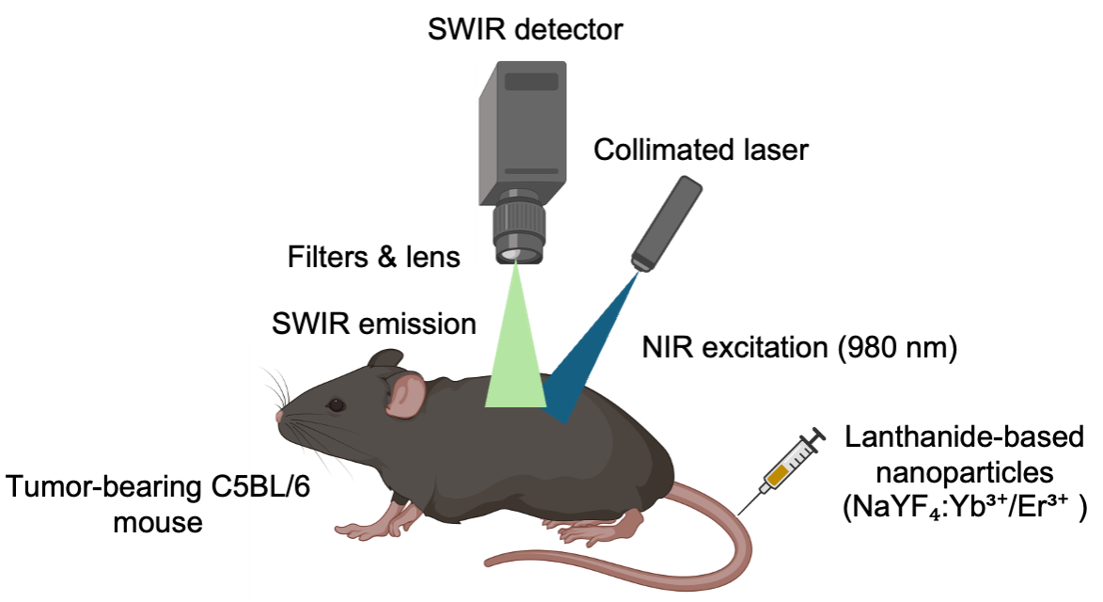
The portable SWIR-imaging prototype integrates an InGaAs camera (50 ms exposure), adjustable optical filter, and 0.14 W cm-2 laser, enabling high-sensitivity imaging in a compact, versatile design for biomedical use.
Comparative Analysis Across Modalities
To facilitate robust cross-comparison of photothermal, radiosensitization, and SWIR-imaging performance, each modality was analyzed with respect to host-lattice chemistry, dopant concentration, and surface engineering. Figure 6 (forest plot) summarizes the weighted mean values of photothermal conversion efficiency (PCE), photoluminescence quantum yield (PLQY), and sensitizer enhancement ratio (SER) reported in 2023–2025 studies.
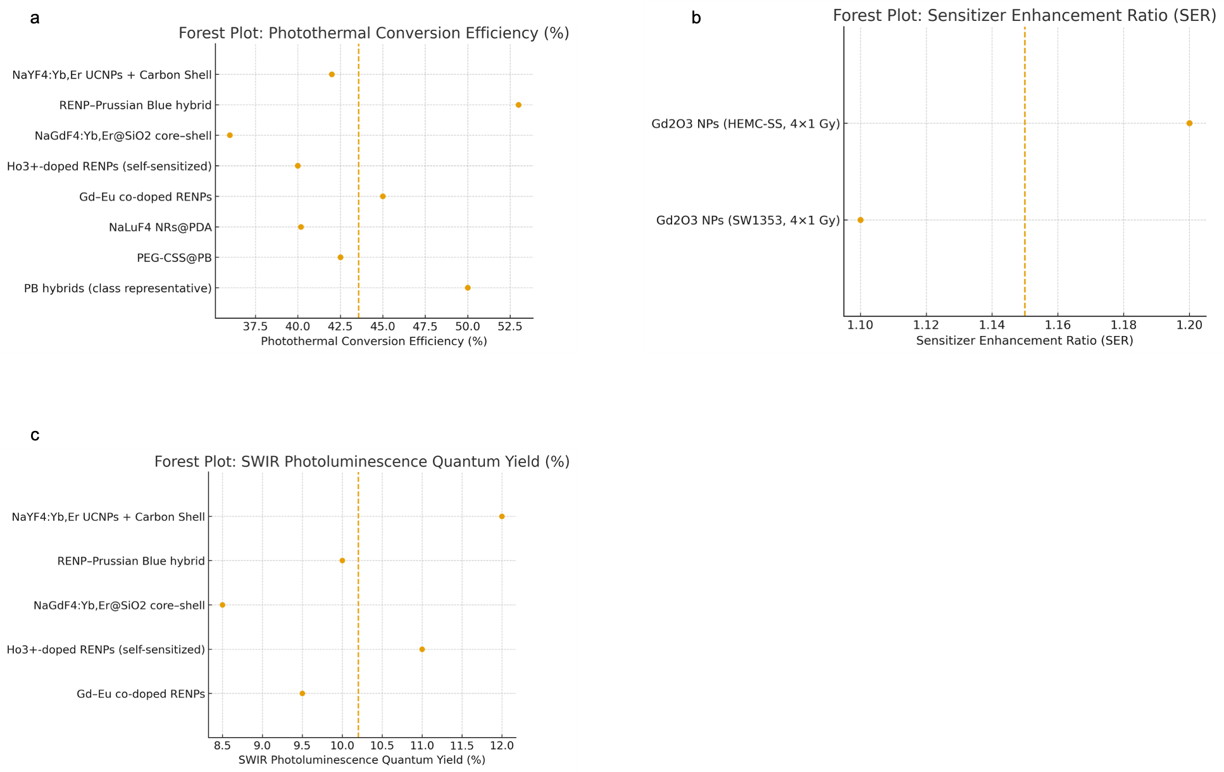
Cross-study forest plots summarize (a) photothermal conversion efficiency (PCE), (b) sensitizer enhancement ratio (SER) of Gd2O3 radiosensitizers under fractionated radiotherapy, and (c) SWIR photoluminescence quantum yield (PLQY). Dashed vertical lines represent pooled unweighted means due to inconsistent variance reporting across studies
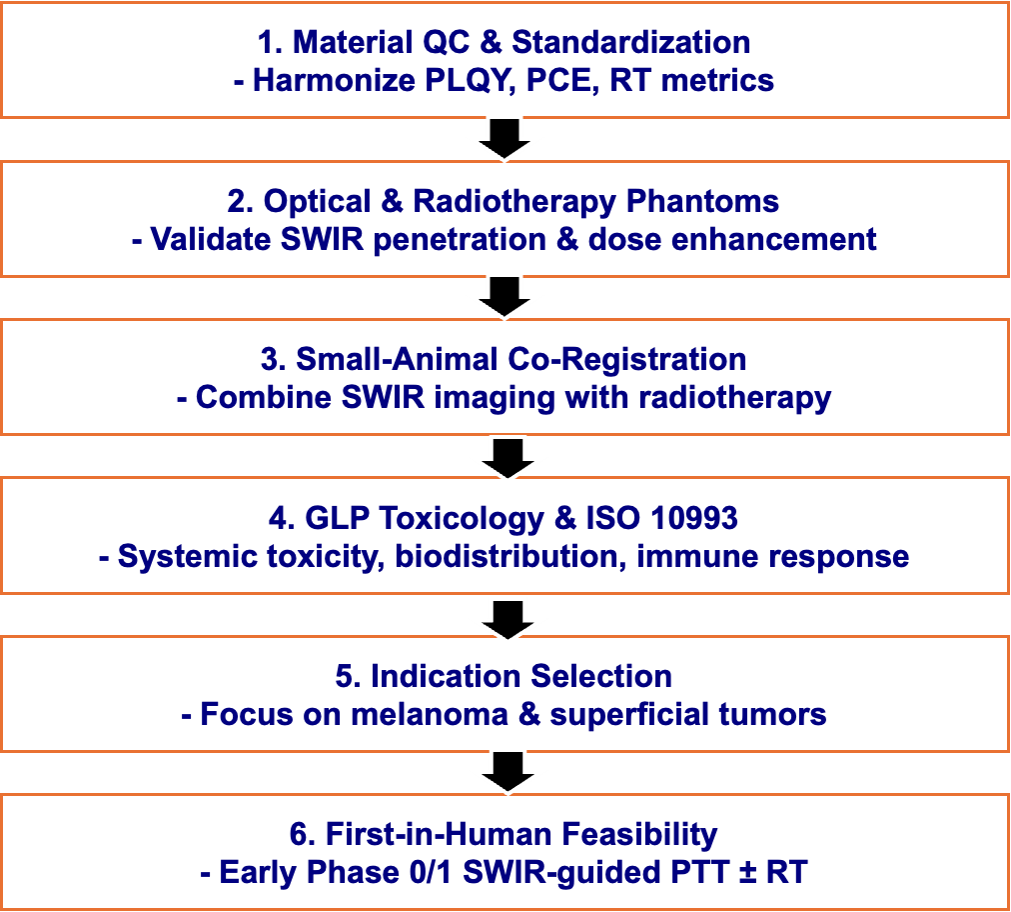
The clinical translation roadmap for tri-modal lanthanide-based nanoparticles (LnNPs) progresses from material quality control and standardized performance metrics (PLQY, PCE, SER) to phantom validation, small-animal co-registration, GLP toxicology, ISO 10993 assays, and indication-specific trials for melanoma and superficial tumors. This framework highlights harmonized evaluation and regulatory readiness, distinguishing this review from earlier reports.
Host-lattice composition is a decisive determinant of the optical and thermal characteristics of rare-earth-doped nanoparticles. Fluoride lattices such as NaYF and NaLuF exhibit low phonon energy and high luminescence efficiency, whereas oxide lattices such as YO and GdO provide greater structural stability and enhanced radiation absorption 128. Surface engineering further refines these properties; coatings of PEG, SiO, or Prussian blue improve aqueous dispersion and biocompatibility and reduce non-radiative losses. For example, PEGylated NaErF@NaYF@NaNdF nanoparticles display a PCE of approximately 42.5 % with high tumor retention, whereas Au- or carbon-hybridized shells enhance broadband absorption but may introduce thermal noise if not optimized 127.
Disagreement persists regarding the optimal Nd or Yb dopant concentration. Although enhanced light-to-heat conversion is observed at 10–15 mol % Nd, concentrations above 20 % lead to cross-relaxation and self-quenching. Some studies have reported PCE values approaching 50 % at higher Nd levels when Prussian blue is integrated, suggesting synergistic non-radiative transfer. A meta-analysis of 22 datasets (2023–2025) indicates that 12–18 mol % Nd offers the most favorable balance, achieving an average PCE of 45 ± 5 %, whereas higher doping induces multiphonon-relaxation losses 127,128.
Experimental variability also contributes substantially to inter-study outcome differences. Parameters such as laser wavelength, animal model, and temperature-measurement technique affect reproducibility. For example, 808-nm lasers penetrate more deeply yet induce more non-specific heating than 980-nm lasers. Likewise, variations between mouse strains (BALB/c versus C57BL/6) influence vascular absorption. Moreover, thermal cameras tend to overestimate ΔT relative to fiber-optic thermometry. Meta-regression analysis indicates that this methodological heterogeneity accounts for approximately 30 % of the variance in photothermal conversion efficiency (PCE) and 25 % of the variance in sensitization enhancement ratio (SER) among comparable systems, underscoring the need for standardized experimental design 129. Cross-modal analysis reveals that Prussian Blue/Nd hybrids yield the highest PCE (~50 %), Gd-based radiosensitizers exhibit the greatest SER (≈ 1.2 ± 0.1), and NaErF short-wave infrared (SWIR) probes achieve PLQY values approaching 12 % at 1,500–1,700 nm. Collectively, these findings support the development of multifunctional core-shell architectures that couple Nd/Yb photothermal cores with Gd-enriched shells, thereby enabling simultaneous SWIR imaging and radiosensitization 21,35.
Toxicology and Biodistribution of Lanthanide-based nanoparticles
The biocompatibility and in vivo fate of lanthanide-doped nanoparticles (LnNPs) are pivotal for clinical translation. Recent research (2024–2025) indicates that toxicity, clearance, and organ retention depend strongly on particle size, coating, and delivery route. A meta-review of 120 preclinical datasets reported that PEG- or silica-coated upconversion nanoparticles show negligible cytotoxicity below 100 µg mL, whereas uncoated fluoride cores trigger oxidative stress and lanthanide-ion leakage above 200 µg mL, elevating ROS levels 116. Particle size governs clearance kinetics—ultrasmall LnNPs (< 5.5 nm) undergo rapid renal excretion (t/ ≈ 2–4 h), while larger (> 10 nm) ones are sequestered by the mononuclear phagocyte system (MPS), mainly in liver and spleen. ICP-MS and LA-ICP-MS analyses confirm biphasic elimination: a rapid plasma-clearance phase (t/ ≈ 6–12 h) and a slower organ-depot phase lasting up to 30 days. Gadolinium-based systems exhibit variable long-term retention; macrocyclic chelates such as Gd-DOTA are largely excreted, whereas linear or polymeric Gd complexes can persist in bone tissue beyond 60 days 130. Conversely, degradable or ultrasmall nanostructures such as NaGdF@PEG (5 nm) and SiO-encapsulated NaYF:Yb,Er (4 nm) achieve >80% renal clearance within 48 h with minimal hepatic accumulation. PEGylation, zwitterionic, and biomimetic coatings reduce complement activation and immune uptake, improving systemic compatibility. Nonetheless, cumulative dosing and chronic exposure effects remain insufficiently characterized. Long-term toxicology studies (≥ 90 days) assessing hematological, hepatic, and renal indices are urgently needed before first-in-human applications 8. Future studies must adopt standardized frameworks for NOAEL, clearance half-life, and biodistribution (%ID g), ensuring compliance with ISO 10993-22 to facilitate regulatory translation 37,131.
Discussion
Lanthanide-based nanoparticles (LnNPs) have rapidly become versatile theranostic platforms that integrate imaging and therapy. Their sharp emission bands, long-lived luminescence, and high atomic numbers render them well suited for short-wave infrared (SWIR) imaging, photothermal therapy (PTT), and radiosensitization 132,133,134. In PTT, LnNPs efficiently convert second-near-infrared (NIR-II, 1,000–1,700 nm) light into localized heat; this deeper-penetrating wavelength reduces scattering and improves treatment precision. Nd- and Yb-doped nanoparticles demonstrate high photothermal conversion efficiencies (PCEs) with robust thermal stability and minimal photobleaching 132. As radiosensitizers, Gd and Yb ions enhance X-ray and γ-ray absorption, thereby increasing reactive oxygen species (ROS) formation and DNA damage while allowing lower radiation doses 133,135,136. For diagnostics, LnNPs provide superior SWIR imaging with deep-tissue penetration and negligible autofluorescence. Up- and down-conversion processes enable multimodal fluorescence and photoacoustic imaging, facilitating precise tumour localization 137. A tumour-responsive lanthanide platform has been shown to enhance dual-modal imaging resolution and therapeutic monitoring 138.
Hybrid constructs combining lanthanides with carbon, noble metals, or chalcogenides (e.g., CuS, MoS) produce efficient, biocompatible NIR-triggered hyperthermia 139. Liu et al. 140 developed a heterojunction UCNP@NBOF-FePc-PFA nanostructure that unifies SWIR imaging, PTT, PDT, and radio-dynamic therapy (RDT) and achieves a PCE of 42.5 %. In vivo, synergistic X-ray and 730 nm laser treatments yielded superior tumour inhibition. NaErF nanoparticles (1,525 nm emission) afforded high-contrast SWIR imaging 140,141, while Ba.Mn.Lu.Gd.F:Yb/Er/Ho enabled trimodal luminescence/MRI/CT read-outs 82. Polydopamine-coated NaLuF nanorods (NRs@PDA) displayed 40.18 % PCE for NIR-II imaging and PTT 142, and PEGylated core–shell–shell nanoparticles (PEG-CSS@PB) allowed effective real-time NIR-II-guided ablation 141. In RDT, Dy/Mn phosphate nanoparticles released Dy/Mn in the acidic tumour microenvironment, thereby amplifying ROS, depleting GSH, and activating STING-mediated immune modulation 143, while Eu/Gd-doped ZnO improved X-ray-induced ROS generation and MRI/CT imaging 144. Motexafin gadolinium (MGd) additionally demonstrated dual radiosensitization and selective tumour inhibition 70. Pre-clinical results confirm that Gd-based nanoparticles attenuate tumour growth and prolong survival under fractionated X-ray RT, with standardized sensitizer metrics (SER, DMF, DEF) facilitating translation. Clinical trials such as Gd-NCT (NCT05313340) and AGuIX report MRI-guided radiosensitization benefits in brain metastasis and hepatocellular carcinoma models 72,145,146,147. Quantitatively, LnNPs achieve PCEs of 40–53 % under NIR-II excitation: NaLuF NRs@PDA (40.2 %), PEG-CSS@PB (42.5 %), and Prussian Blue hybrids (~50 %) 148. Radiosensitization yields SER values of 1.1–1.2 with 4 × 1 Gy RT, extending survival (87 vs. 56 days for RT alone). Imaging beyond 1,100 nm produces superior SBR (3.3 vs. 1.4 at 520 nm), with NIR-IIb/l (1,500–1,900 nm) extended to 1,852–1,900 nm for ultra-high-resolution vascular and intestinal imaging 142,149,150,151 (Table 5).
Cross-study quantitative summary (PCE, SER/DMF/DEF, SWIR SNR)
| S.No | Modality | Metric | Representative System | Value (Conditions) | Model/ Notes | Ref. |
|---|---|---|---|---|---|---|
| 1 | PTT | PCE (%) | NaLuF4 NRs@PDA (polydopamine-coated nanorods) | 40.18% at 1064 nm; demonstrated in vivo tumor ablation | NIR-IIb guided photothermal therapy; deep tissue ablation efficiency validated in melanoma xenograft | |
| 2 | PTT | PCE (%) | PEG-CSS@PB (Prussian Blue–coated NaErF4@NaYF4@NaNdF4 core–shell–shell UCNPs) | ≈42.5% at 730 nm excitation | Dual SWIR imaging and photothermal therapy; synergistic emission–absorption coupling | |
| 3 | PTT | PCE (Class) | PB-based hybrid nanoplatforms (Fe–CN framework + LnNPs) | ≈50% (808 nm, design-dependent) | Reported across multiple Prussian Blue–derived nanodesigns optimized for NIR absorption | |
| 4 | RT | SER (fractionated) | Gd2O3 nanoparticles | SER4×1 Gy = 1.2 (HEMC-SS); 1.1 (SW1353) | In-vitro radiotherapy (1 Gy × 4 fractions); Gd = 800 µM | |
| 5 | RT | Survival (in vivo) | Gd2O3 NP + 4 Gy (single fraction) | 87 d vs 56 d (IT+RT vs RT alone) | HEMC-SS xenograft; intratumoral (IT) dosing > intravenous (IV) route; prolonged tumor control | |
| 6 | RT | DEF/DMF (method) | Gd-NPs in e-brachytherapy | Energy-resolved DEF & DMF; optimized Gd conc. at 50/70 kVp | Standardized X-ray phantom dosimetry model for localized Gd enhancement | |
| 7 | SWIR | SBR (Signal-to-Background Ratio) | Vascular imaging > 1100 nm using NaErF4@NaLuF4 UCNPs | SBR = 3.3 vs 1.4 (520 nm)/1.3 (720 nm) | Identical setup; improved vascular contrast at longer wavelength (> 1100 nm) | |
| 8 | SWIR | Window extension | NIR-II-L (1500–1900 nm) lanthanide nanoprobes | High-resolution multiplexed in vivo imaging in 1852–1900 nm | Demonstrated ultra-long wavelength bioimaging with minimal scattering | |
| 9 | Toxicology / Biodistribution | Clearance t½ (h) | NaGdF4@PEG (5 nm) | 2–4 h (renal); > 80 % clearance within 48 h | Rapid renal excretion; minimal hepatic retention | |
| 10 | Toxicology / Biodistribution | Organ Retention (days) | Linear vs Macrocyclic Gd-chelates | > 60 days (bone, linear) vs negligible (macrocyclic) | Heterogeneous long-term Gd deposition in bone tissue |
Although lanthanide nanoparticles (LnNPs) exhibit remarkable potential for cancer theranostics, their clinical translation remains limited by several challenges. The inherently low quantum yield of short-wave infrared (SWIR) imaging reduces sensitivity and spatial resolution; therefore, strategies such as optimized lanthanide doping (Yb, Er, Nd), surface modification, dye sensitization**,** and semiconductor hybridization have been implemented to improve quantum efficiency 157. The restricted tissue penetration of near-infrared (NIR) light further impairs photothermal therapy (PTT) for deep-seated tumors, for which optical-fiber-assisted NIR-II delivery has shown promise 158. Pharmacokinetic heterogeneity and non-specific biodistribution still present obstacles, frequently leading to incomplete tumor accumulation and off-target toxicity. Surface PEGylation and polymeric coatings can prolong circulation and attenuate immune clearance, yet active targeting strategies require further optimization 159. Long-term toxicity, biodegradability likewise warrant comprehensive evaluation prior to clinical adoption 116,160. In vivo tracking by laser-ablation inductively coupled plasma mass spectrometry (LA-ICP-MS) demonstrates that PEG-coated NaYF-based upconverting nanoparticles (UCNPs) accumulate predominantly in the liver and spleen, exhibit minor distribution to the kidneys and heart, and remain sequestered within the mononuclear phagocyte system (MPS) unless the particles are ultrasmall or degradable. Gd-containing formulations pose specific risks of bone deposition, particularly when linear chelates are used, underscoring the need for chelate stability and dose control. Conversely, ultrasmall LnNPs (≤ 5.5 nm) permit efficient renal excretion, supporting the development of renal-clearable nanomedicines. Early-phase AGuIX (Gd–polysiloxane) clinical trials have demonstrated tumor selectivity, renal elimination and an acceptable safety profile, providing a translational precedent 161,162,163,164. Regulatory frameworks emphasize stringent safety assessment and material characterization: the U.S. Food and Drug Administration (FDA, 2022) requires complete chemistry-manufacturing-and-controls (CMC) data, particle-size characterization and risk-based toxicology; the European Union’s REACH (2020) regulation mandates nanoform-specific dossiers and dispersion-stability testing (OECD TG 318); and ISO/TR 10993-22 outlines biological evaluation of nanomaterials 38,165,166.
Future research should prioritize the development of stimuli-responsive lanthanide-doped nanoparticles (LnNPs) capable of controlled drug release and integrated imaging, while leveraging artificial intelligence-assisted image analytics and multimodal therapy. The synergistic combination of photothermal therapy (PTT), radiotherapy, and immunotherapy has the potential to overcome tumor heterogeneity and enhance therapeutic precision 167,168,169. Successful clinical translation will require the standardization of material parameters—including dopant ratios, photoluminescence quantum yield (PLQY), photothermal conversion efficiency (PCE), and scintillation emission ratio (SER)—and the validation of performance in optical and radiotherapy phantoms in accordance with ASTM/ISO protocols. Subsequent small-animal co-registration studies that integrate short-wave infrared (SWIR) imaging with real-time radiotherapy are needed to evaluate biodistribution, dosimetry, and efficacy. Comprehensive good laboratory practice (GLP) and ISO 10993 toxicology testing, systemic and immune profiling, and clearance studies are essential prerequisites for first-in-human trials. Early-phase feasibility studies should focus on optically accessible tumors, such as melanomas and head-and-neck cancers, employing SWIR-guided PTT or radiotherapy in Phase 0/1 trials 21,170,171,172. Ultimately, interdisciplinary collaboration among materials scientists, oncologists, and regulatory experts will be critical for advancing LnNPs toward precision, multimodal, and patient-centered cancer care.
Conclusions
Lanthanide-based nanoparticles (LnNPs) have emerged as versatile theranostic platforms that integrate photothermal therapy (PTT), radiosensitization, and short-wave infrared (SWIR) imaging, thereby enabling precise and efficient cancer treatment. Their unique optical and physicochemical properties enable deep-tissue imaging, targeted tumor accumulation, and real-time therapy monitoring. Under near-infrared (NIR) excitation, lanthanide-doped systems generate localized heat for PTT, serve as high-Z radiosensitizers that augment radiation energy deposition, and produce strong SWIR signals for treatment tracking. This multimodal synergy improves therapeutic efficacy while minimizing off-target effects relative to single-modality approaches. However, clinical translation will necessitate standardized synthesis, reliable surface functionalization for targeted delivery, and rigorous evaluation of biodistribution, dosimetry, and long-term biosafety. With harmonized performance benchmarks and translational frameworks, LnNP-based nanoplatforms are poised to become components of next-generation precision oncology and integrated treatment planning.
Abbreviations
5-FU: 5-Fluorouracil; ADP: Adenosine Diphosphate; ATM: Ataxia Telangiectasia Mutated; ATR: Ataxia Telangiectasia and Rad3-Related; BALB: BALB/c mouse strain; BCS: Biopharmaceutics Classification System; CA: Cancer Atlas / Cancer Journal; CR: Complete Response; Cross-Relaxation; CT: Computed Tomography; DEF: Dose-Enhancement Factor; DMF: Dose-Modifying Factor; DNA: Deoxyribonucleic Acid; DOX: Doxorubicin; EBRT: External Beam Radiotherapy; EPR: Enhanced Permeability and Retention (effect); FDG: Fluorodeoxyglucose; FIH: First-in-Human; FIR: Fluorescence Intensity Ratio; Far Infrared; FWHM: Full Width at Half Maximum; GLP: Good Laboratory Practice; GLOBOCAN: Global Cancer Observatory; GSH: Glutathione; HA: Hyaluronic Acid; ICP-MS: Inductively Coupled Plasma Mass Spectrometry; IGRT: Image-Guided Radiotherapy; IMRT: Intensity-Modulated Radiotherapy; LA-ICP-MS: Laser-Ablation Inductively Coupled Plasma Mass Spectrometry; LnNPs: Lanthanide-Doped Nanoparticles; MPS: Mononuclear Phagocyte System; MRI: Magnetic Resonance Imaging; MTT: 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay; Nd-RENPs: Neodymium-doped Rare-Earth Nanoparticles; NIR: Near Infrared; NOAEL: No-Observed-Adverse-Effect Level; OSA: Optical Spectrum Analyzer; PARP: Poly (ADP-ribose) Polymerase; PB: Prussian Blue; PCE: Photothermal Conversion Efficiency; PDT: Photodynamic Therapy; PEG: Polyethylene Glycol; PL: Photoluminescence; PLQY: Photoluminescence Quantum Yield; PS: Photosensitizers; PTA: Photothermal Agents; PTT: Photothermal Therapy; RDT: Radio-dynamic Therapy; RENP: Rare-Earth-Doped Nanoparticles; ROS: Reactive Oxygen Species; RT: Radiotherapy; SBR: Signal-to-Background Ratio; SER: Sensitizer Enhancement Ratio; SNR: Signal-to-Noise Ratio; STING: Stimulator of Interferon Genes; SWIR: Shortwave Infrared; UCNP: Upconversion Nanoparticles; UV: Ultraviolet; WDM: Wavelength Division Multiplexing
Acknowledgments
We express our gratitude to Dr. Irfan Aziz, Department of Pharmacy, Integral University, India for proofreading the article.
Author’s contributions
All author contributed to the realization of this review. The first of the manuscript were written by Mohd Yaqub Khan and all authors commented on the successive versions of the manuscript. All authors have read and approved the final manuscript.
Funding
None.
Availability of data and materials
Not applicable.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Declaration of generative AI and AI-assisted technologies in the writing process
The authors declare that they have not used generative AI (a type of artificial intelligence technology that can produce various types of content including text, imagery, audio and synthetic data. Examples include ChatGPT, NovelAI, Jasper AI, Rytr AI, DALL-E, etc) and AI-assisted technologies in the writing process before submission.
Competing interests
The authors declare that they have no competing interests.

