Impact of patulin on apoptosis, autophagy, and tumor growth in human malignant mesothelioma and normal mesothelial cells
- Department of Pharmaceutical Toxicology, Faculty of Pharmacy, İnönü University, Malatya, Turkey
- Department of Pharmaceutical Toxicology, Faculty of Pharmacy, Fırat University, Elazığ, Turkey
- Department of Biophysics, Faculty of Medicine, Pamukkale University, Denizli, Turkey
Abstract
Patulin (PAT) is a mycotoxin produced by certain molds, particularly species of the genera Aspergillus and Penicillium. It is found predominantly in apple-derived products and is associated with both acute and chronic toxicities. Although comprehensive data on the anti-cancer properties of PAT are presently lacking, preliminary evidence suggests that it can influence cancer cells by enhancing cellular apoptosis and inducing cytotoxicity. To evaluate the potential of PAT as a therapeutic approach for malignant mesothelioma, we investigated its effects on the malignant mesothelioma cell line H2452 and the non-malignant mesothelial cell line MeT-5A. Cells were exposed to PAT (1–200 µM). Cell viability was assessed using the MTS assay, and cell migration was evaluated using the wound-healing assay. Expression levels of genes related to autophagy and apoptosis (SQSTM1, Beclin-1, Atg5, Atg7, Bax, Bcl-2) were measured using RT-PCR. PAT produced a dose-dependent decrease in cell viability and exhibited greater cytotoxicity toward mesothelioma cells than toward healthy cells. The wound-healing assay demonstrated that PAT impeded cellular migration, as evidenced by diminished wound closure. In cancer cells, PAT down-regulated Bax, Bcl-2, Beclin-1, and SQSTM1, while up-regulating Atg5 and Atg7. In healthy cells, it reduced Bax, Bcl-2, Beclin-1, SQSTM1, and Atg7, and elevated Atg5. It diminished apoptosis in both healthy and cancer cells while modulating autophagy pathways in both cell types, potentially attenuating autophagy in cancer cells and disrupting this process in healthy cells. Our findings indicate that PAT may significantly restrict tumor progression in human mesothelial cancer models.
Introduction
Mycotoxins are secondary metabolites produced by fungi, and more than 500 distinct compounds have been identified. The main producers of these toxins are molds belonging principally to the genera Penicillium, Aspergillus, Alternaria, Fusarium, and Claviceps. Despite their small molecular size and remarkable stability, mycotoxins can be highly hazardous in foods 1,2. The mycotoxin PAT (4-hydroxy-4H-furo[3,2-c]pyran-2(6H)-one, C7H6O4) is produced predominantly by Penicillium, Aspergillus, and Byssochlamys species. PAT is a lactone composed of a furan ring conjugated to a β-diketone structure that contains two carbonyl groups (Figure 1). High concentrations of PAT are found in apple-based products, especially apple jam, apple juice, and apple wine 1,3.
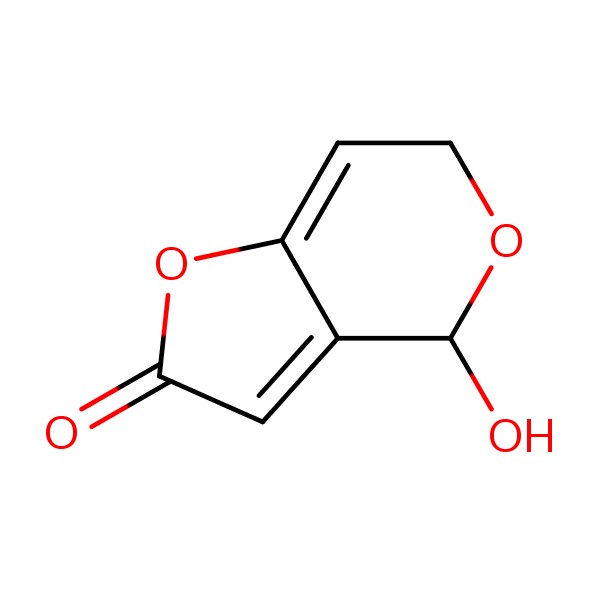
Structure of patulin (PAT).
Exposure to PAT may occur via ingestion of contaminated food, inhalation of contaminated dust during food processing, or through direct dermal contact. Dietary intake of PAT depends on the type as well as the amount of fruit and fruit-derived products consumed 3. The World Health Organization (WHO) has established a maximum allowable PAT concentration of 50 µg kg in apple juice 4. PAT can cause both acute and chronic toxic effects. Acute toxicity is characterized by gastrointestinal inflammation, ulcers, nausea, vomiting, diarrhea, gastrointestinal bleeding, and cerebral edema; in severe cases, hepatic injury may ensue. Multiple studies suggest that exposure to varying concentrations of PAT can elicit genotoxic, teratogenic, embryotoxic, and immunotoxic effects in animals and certain microorganisms 1. The International Agency for Research on Cancer (IARC) has classified PAT in Group 3 as ‘not classifiable as to its carcinogenicity to humans’ 5.
Given the limitations of conventional anticancer therapies, researchers are exploring novel compounds and formulations that may provide greater efficacy with fewer adverse effects 6. In this context, toxins and metabolites produced by bacteria and fungi are being evaluated as potential therapeutic agents 7. PAT exerts its toxicity through two principal mechanisms: (i) induction of oxidative stress and (ii) covalent binding to reactive sulfhydryl groups in amino acids, glutathione, and cellular proteins 8. Numerous toxicological studies have demonstrated that oxidative stress is a key pathway by which PAT triggers apoptosis 9. It is therefore essential to determine whether oxidative damage also influences other pathways and to clarify the mechanisms underlying PAT-induced cytotoxicity. Excessive generation of reactive oxygen species (ROS) can provoke extensive cellular injury, including oxidative DNA damage and activation of cell-cycle checkpoints, ultimately leading to cell-cycle arrest 10. Although PAT is recognized for targeting thiol groups and promoting ROS production, the precise mode of cell death in cultures exposed to PAT remains unresolved. The present study investigates, for the first time, in mesothelioma (H2452) and non-malignant mesothelial (MeT-5A) cells, how PAT treatment modulates apoptotic and autophagic pathways.
Malignant mesothelioma (MM) is a rare, highly aggressive neoplasm that most commonly arises in the pleura and is predominantly associated with inhalational exposure to asbestos or erionite fibers. Approximately 80 % of cases originate in the pleura and are referred to as malignant pleural mesothelioma (MPM) 11. The disease typically exhibits an indolent onset and is therefore frequently diagnosed at an advanced stage, because contemporary imaging modalities lack sufficient sensitivity for early detection and no reliable serum biomarker has yet been validated. In addition, the anatomical configuration of the serosal cavities facilitates rapid dissemination and invasion of malignant cells. Pathologically, MM is categorized into three major histological subtypes—epithelioid, sarcomatoid, and biphasic—the latter containing both epithelioid and sarcomatoid components 12.
A principal objective of anticancer drug development is the identification of actionable molecular targets that exploit tumor-specific vulnerabilities. Accumulating evidence indicates that enhanced autophagic flux constitutes a key survival mechanism in cancer cells, thereby nominating autophagy as a therapeutic target. The multidomain protein p62/sequestosome-1 (SQSTM1) serves as an autophagic cargo adaptor: it binds LC3, promotes autophagosome formation, and mediates the sequestration of ubiquitinated proteins into inclusion bodies. Intracellular accumulation of p62 has been linked to tumorigenesis, whereas autophagy suppresses malignant transformation by limiting p62 levels 13. Additional regulators critical for the nucleation phase include Beclin-1 (BECN-1) and the anti-apoptotic protein Bcl-2; binding of Bcl-2 to BECN-1 inhibits autophagy. Over-expression of Bcl-2 under metabolic stress impairs BECN-1 function and thereby suppresses autophagy 14.
The present study investigated the effects of increasing concentrations of PAT on cell viability, migration, and the expression of selected autophagy-related (SQSTM1, Beclin-1, ATG5, ATG7) and apoptosis-related (Bax, Bcl-2) genes in MeT-5A and H2452 cells. The results demonstrate that PAT modulates both autophagic and apoptotic pathways, suggesting its potential utility as a therapeutic agent for malignant mesothelioma.
Materials and methods
Chemicals and cell culture conditions
PAT was purchased and dissolved in DMSO. MeT-5A and H2452 cells were cultured in DMEM supplemented with 10 % FBS and 1 % penicillin/streptomycin. Cells were maintained at 37 °C in a humidified incubator with 5 % CO and were passaged two to three times per week. Following centrifugation at 1,500 rpm for 5 min, the cell pellet was resuspended in fresh medium to the desired concentration. Stock solutions of PAT were serially diluted to final concentrations of 1, 2.5, 5, 10, 25, 50, 100, and 200 µM in fresh medium. Medium containing 0.1 % DMSO served as the vehicle control (see Table 1 for suppliers and catalogue numbers).
Reagents, suppliers, and catalog numbers used in this study.
| Reagent | Supplier | Catalog Number |
|---|---|---|
| PAT | Cayman Chemicals, USA | 704004 |
| DMSO (Dimethyl sulfoxide) | Sigma-Aldrich, USA | D8418 |
| DMEM (Dulbecco’s Modified Eagle Medium) | Sigma-Aldrich, USA | D6429 |
| Fetal Bovine Serum (FBS) | Gibco, USA | 26140079 |
| Penicillin-Streptomycin (10,000 U/mL) | Gibco, USA | 15140122 |
| Trypsin-EDTA (0.25%) | Gibco, USA | 25200056 |
Cell viability assay
Cell viability was evaluated using the MTS assay. When cultures reached approximately 90 % confluence, cells were harvested with trypsin-EDTA, counted, and seeded into 96-well plates at 5,000 cells/well 24 h before treatment. After overnight incubation (37 °C, 5 % CO), the medium was replaced with PAT-containing medium and the cells were incubated for 24 or 48 h. Absorbance was recorded at 450 nm with a microplate reader (BioTek Instruments, Winooski, VT, USA). The half-maximal inhibitory concentration (IC) was calculated for each cell line using GraphPad Prism 10 software 15. All experiments were performed in quadruplicate.
Wound-healing assay
Cell migration was assessed using a scratch (wound-healing) assay. Briefly, 1 × 10 cells were seeded in 1 ml of medium in 12-well plates. After 24 h of standard incubation (37 °C, 5 % CO), a linear scratch was created across the cell monolayer with a sterile 200 µl pipette tip. The wells were gently washed with medium to remove detached cells, after which fresh medium containing PAT at the previously determined IC concentration was added. Wound closure was photographed at 0, 24, and 48 h using an inverted light microscope (SOPTOP ICX41, China) 16. All experiments were conducted in triplicate.
Autophagic and apoptotic cell death
Primers for SQSTM1, Beclin-1, Atg5, Atg7, Bax, and Bcl-2 were designed using Primer-BLAST (Bethesda, MD, USA) and synthesized by Sentebiolab (Ankara, Turkey). To examine gene expression related to autophagy and apoptosis, 1 × 10 cells were seeded in each well of sterile 6-well plates and allowed to adhere overnight before treatment with PAT at its IC concentration 17. Total RNA was isolated with Trizol reagent (#T9424, Sigma, USA) according to the manufacturer’s instructions. RNA purity and concentration were verified spectrophotometrically. First-strand cDNA was synthesized from 1 µg of total RNA using a commercial kit (Bio-Rad, USA) 18 in a final volume of 20 µL. The reaction was incubated at 55 °C for 15 min followed by 85 °C for 5 min. Synthesized cDNA was stored at –20 °C until analysis. All procedures were performed in triplicate.
Real-time PCR analysis
Real-time PCR was performed on a StepOnePlus system (Applied Biosystems, USA). Target genes involved in autophagy and apoptosis were amplified, with β-actin serving as the endogenous control. Reactions were prepared with 2× SYBR Green Master Mix, dispensed into 96-well plates, and run in triplicate. Primer pairs were added to each well, and nuclease-free water was used to adjust the final volume to 20 µL. Cycling and melt-curve analyses were executed according to the kit protocol. Relative mRNA levels were calculated with GeneGlobe Data Analysis software (Qiagen) using the 2^–ΔΔCT method (Table 2) 19. Experiments were independently repeated three times.
Primer sequences were used in the gene expression analysis.
| Gene | Primer sequence |
|---|---|
| F: CACCATTGGCAATGAGCGGTTC | |
| R: AGGTCTTTGCGGATGTCCACGT | |
| F: TCAGGATGCGTCCACCAAGAAG | |
| R: TGTGTCCACGGCGGCAATCATC | |
| F: ATCGCCCTGTGGATGACTGAGT | |
| R: GCCAGGAGAAATCAAACAGAGGC | |
| F: TGTGTAGCGTCTGCGAGGGAAA | |
| R: AGTGTCCGTGTTTCACCTTCCG | |
| F: CTGGACACTCAGCTCAACGTCA | |
| R: CTCTAGTGCCAGCTCCTTTAGC | |
| F: GCAGATGGACAGTTGCACACAC | |
| R: GAGGTGTTTCCAACATTGGCTCA | |
| F: CGTTGCCCACAGCATCATCTTC | |
| R: CACTGAGGTTCACCATCCTTGG |
Statistical analysis
Cell-viability and wound-healing data are expressed as percentages. Wound closure was quantified with ImageJ. Statistical analyses were conducted in SPSS v22.0 (SPSS Inc., Chicago, IL, USA). Two-way ANOVA followed by Bonferroni post-hoc testing was applied, and differences were considered significant at p < 0.05. IC values were calculated with GraphPad Prism v10.
Results
Impact of PAT on cellular viability
Cell lines were exposed to PAT for 24 or 48 h to assess its inhibitory effect on cell viability. Cell viability was reported as the IC50 values and proliferation rate (Figure 2). The MTS assay demonstrated that PAT induces cytotoxic effects in MeT-5A and H2452 cell lines. The IC50 values for MeT-5A and H2452 cell lines were 101.5 µM and 12.68 µM, respectively, after 24 h of incubation, and 62.95 µM and 29.64 µM, respectively, after 48 h of incubation (Figure 2). Collectively, these data indicate that H2452 malignant cells are considerably more sensitive to PAT than non-malignant MeT-5A cells.
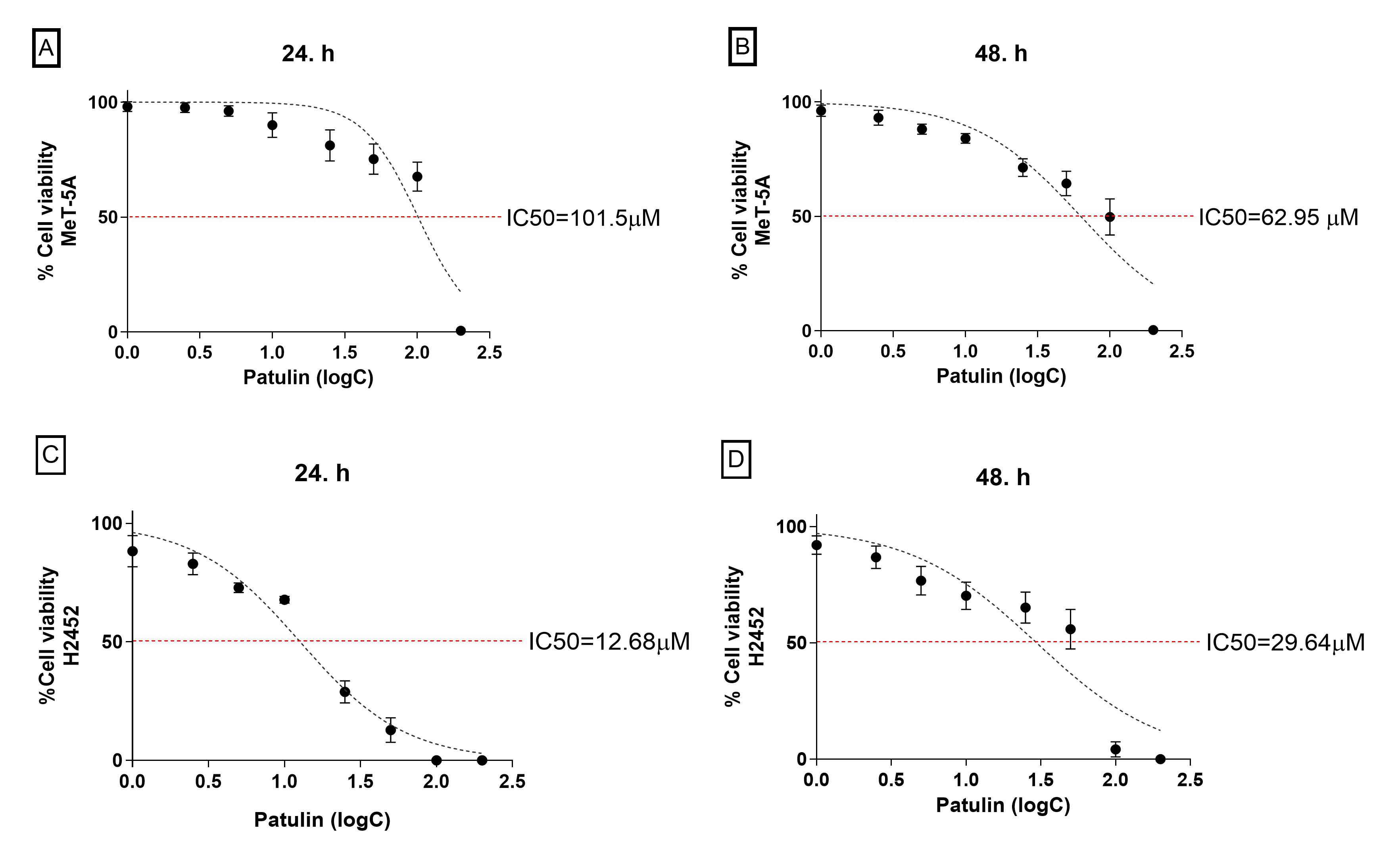
MTS [3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium] cell viability assay. Changes in cell viability percentages of MeT-5A (A, B) and H2452 (C, D) cell lines treated with increasing concentrations of patulin (PAT) (1-200 µM) for 24 and 48 hours.
Effect of PAT on cell migration
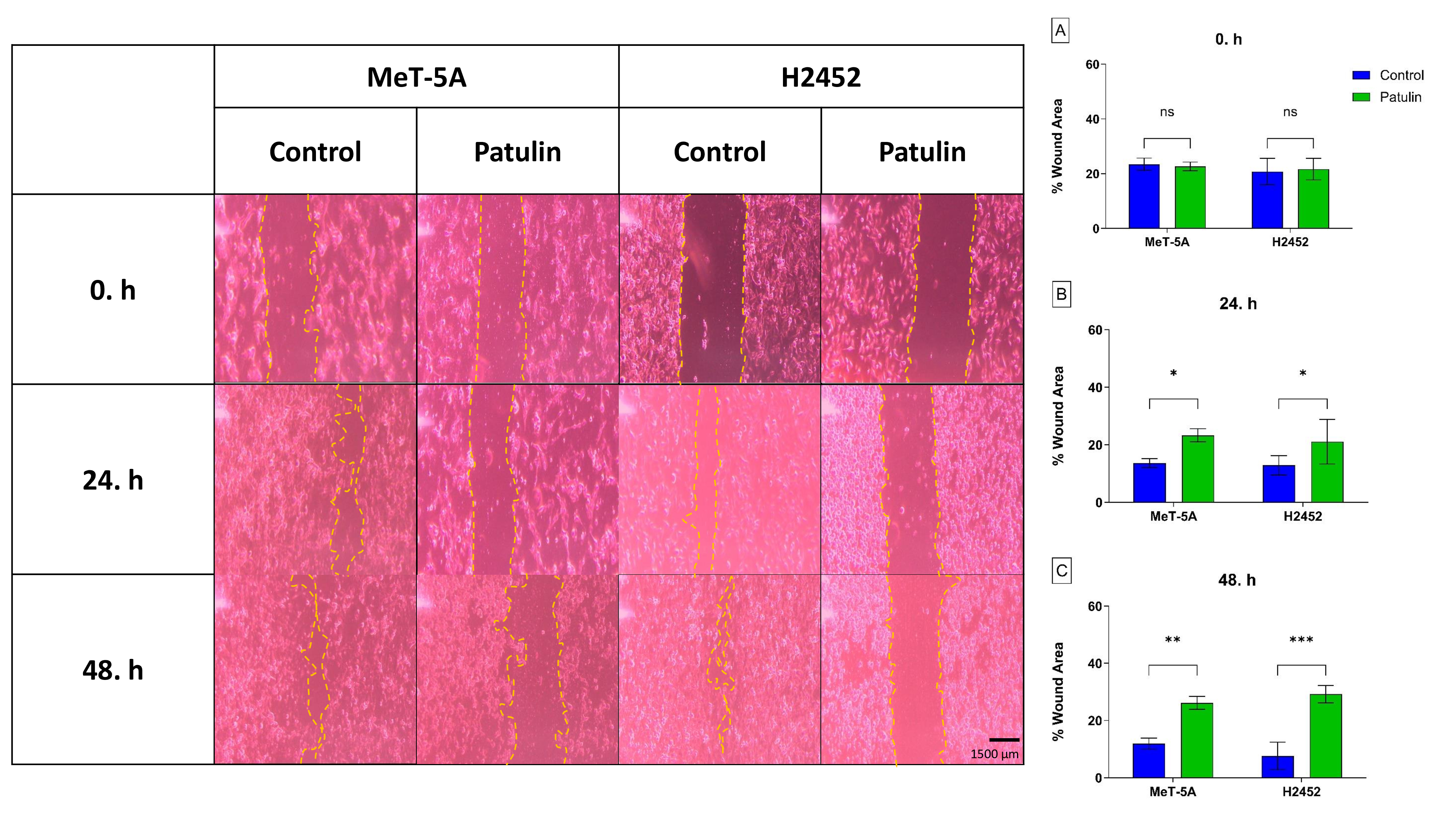
The wound-healing assay demonstrated that PAT treatment altered cell motility, resulting in a modulation of in vitro wound closure rather than a clear acceleration, consistent with the low migration rate observed during the first 24–48 h (Figure 3 and Table 3). These results suggest an influence on cellular migration under the assay conditions; however, they do not necessarily indicate protumorigenic activity, which must be verified in additional invasion and metastasis models.
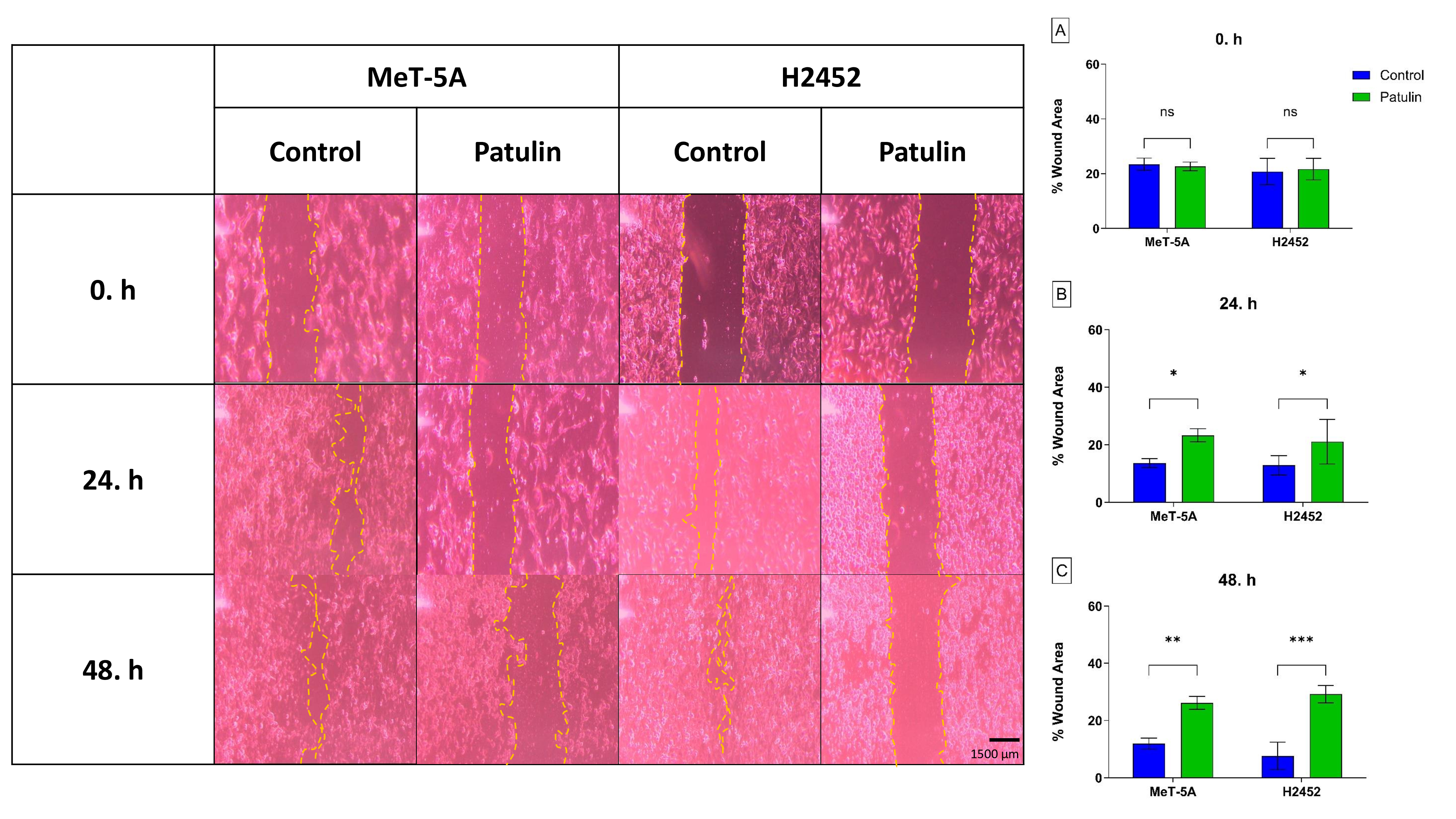
Changes in the wound area of patulin (PAT)-treated mesothelial cells over time. The percentage wound area of control cells and cells treated with PAT IC50~ concentration was measured at 0, 24, and 48 hours in MeT-5A and H2452 cell lines. *p<0.05, **p<0.01, ***p<0.001 compared to control (Scale bar = 1500 µm, Magnification = 40X).
Wound area percentages of control cells and patulin (PAT) half-maximal inhibitory concentration (IC50)-treated cells of MeT-5A and H2452 cell lines at 0, 24 and 48 hours. *p<0.05, **p<0.01, ***p<0.001 compared to the control.
| % Wound area | ||||
|---|---|---|---|---|
| Cell line | Group | 0. h | 24. h | 48. h |
| MeT-5A | Control | 23.48 ± 2.26 | 13.67 ± 1.53 | 11.95 ± 1.95 |
| Patulin | 22.69 ± 1.57 | 23.33 ± 2.31* | 26.19 ± 2.27** | |
| H2452 | Control | 20.81 ± 4.82 | 12.86 ± 3.35 | 7.64 ± 4.77 |
| Patulin | 21.73 ± 3.96 | 21.13 ± 7.73* | 29.22 ± 2.30*** | |
Gene expression analysis
The effects of PAT treatment on gene expression in H2452 cells are illustrated in Figure 4. PAT treatment altered the expression of apoptosis- and autophagy-related genes. Relative to the control group, the mRNA levels of the pro-apoptotic protein Bax and the anti-apoptotic protein Bcl-2 were both reduced. Conversely, Atg5 and Atg7 were up-regulated, whereas Beclin-1 and SQSTM1 were down-regulated, indicating a complex impact on autophagy-related gene regulation. Collectively, these findings suggest that PAT modulates the balance between apoptotic and autophagic pathways in H2452 cells; however, functional confirmation at the protein level and evaluation of autophagic flux remain necessary. In MeT-5A cells, PAT produced a comparable transcriptional profile, lowering Bax, Bcl-2, Beclin-1, SQSTM1, and Atg7 while increasing Atg5. These results point to a partial modulation of autophagy and apoptosis, but further studies are required to substantiate functional effects.
Figure 5 illustrates the gene-expression profile of MeT-5A cells after PAT treatment. Expression levels of Bax, Bcl-2, Beclin-1, SQSTM1, and Atg7, all of which are associated with autophagic cell death, were reduced relative to those of the control group. Conversely, Atg5 expression was elevated. These findings indicate that PAT modulates both apoptotic and autophagic pathways, possibly inhibiting critical steps of autophagy while simultaneously influencing apoptosis-related signalling. Together, the data imply a partial modulation of autophagy and apoptosis, effects that appear more pronounced in cancer cells than in normal counterparts, consistent with the IC values.
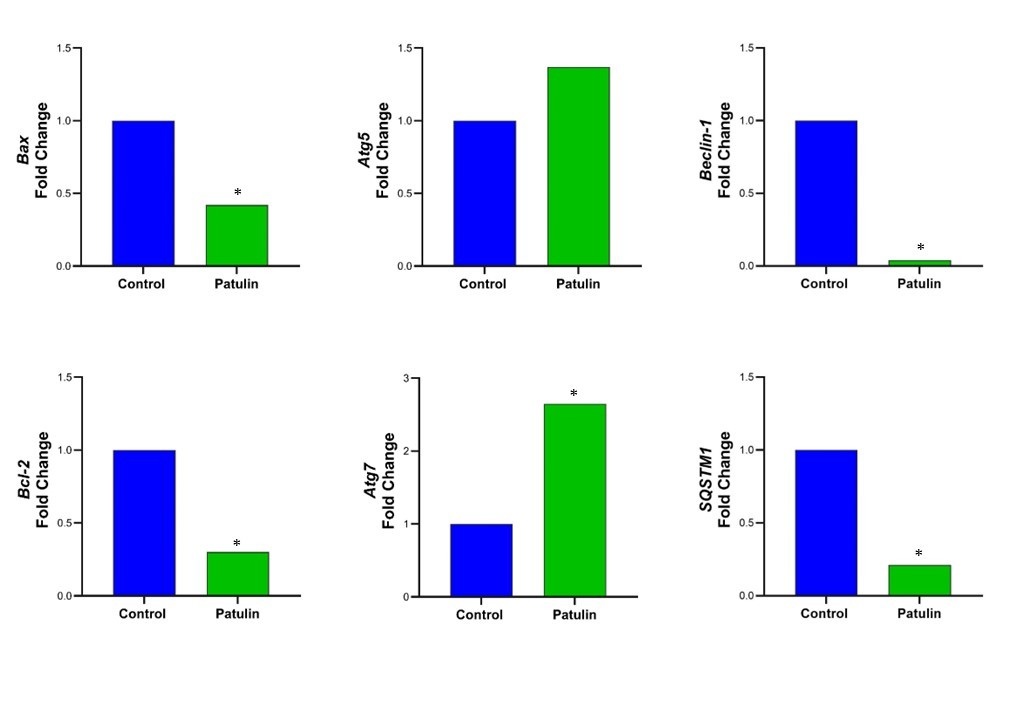
Effects of patulin (PAT) on SQSTM1, Beclin-1, Atg5, Atg7, Bax, Bcl-2 gene expressions in H2452 cells. Gene expression levels were analyzed following treatment with PAT. Data are presented as relative expressions compared to the control. (*p < 0.05).
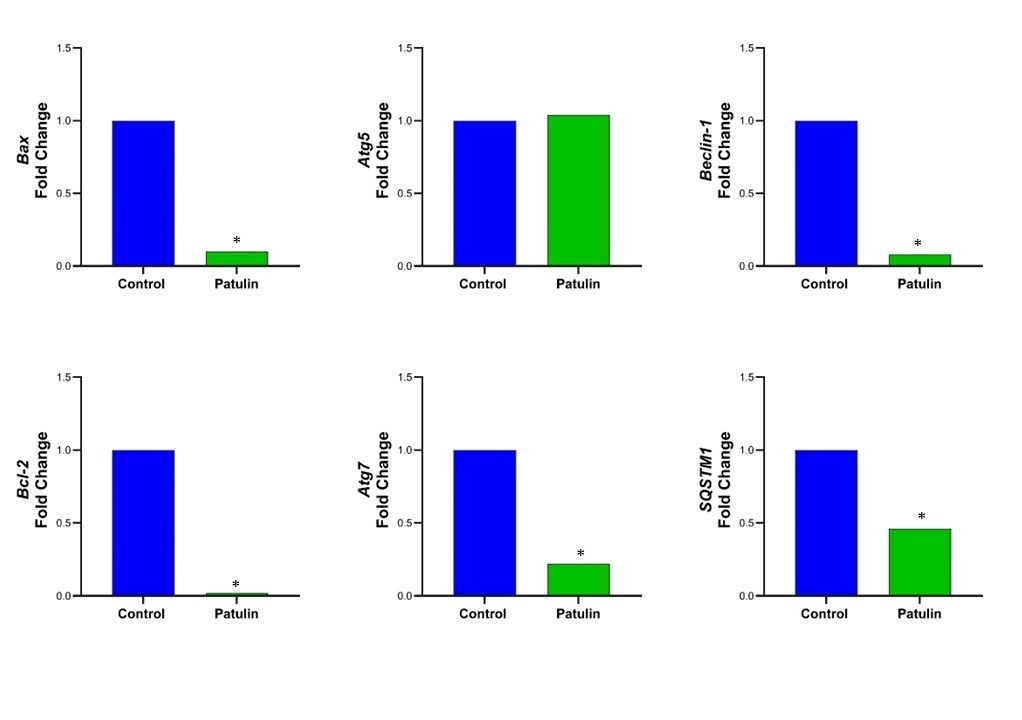
Effects of patulin (PAT) on SQSTM1, Beclin-1, Atg5, Atg7, Bax, Bcl-2 gene expressions in MeT-5A cells. Gene expression levels were analyzed following treatment with PAT. Data are presented as relative expressions compared to the control. (*p < 0.05).
Discussion
PAT is a mycotoxin produced by several species of the genera Aspergillus, Penicillium, and Byssochlamys. PAT is predominantly found in apples, pears, and grapes. Although PAT is reported to possess anticancer activity, its exact biological target remains unclear 7. Only a limited number of studies have investigated the impact of PAT on human cancer cells. Nevertheless, multiple investigations have demonstrated that PAT can trigger apoptosis in various cell types (Table 4) 8,20,21.
In vitro studies on patulin (PAT) related to apoptosis and autophagy were reported in the literature in the last ten years
| Cell line | Dose Time | Results | Ref. |
|---|---|---|---|
| HeLa, SW-48, and MRC-5 | 0.5, 1, 2, and 4 µM of PAT for 24 h | Significant reduction in apoptosis induction in a dose-dependent manner. | 20 |
| HEK293 | 5-15 µM of PAT for 8 h | Oxidative stress plays a key role in PAT-induced apoptosis. | 21 |
| H9C2 | 0-40 µM of PAT for 24 h |
PAT treatment increased the expression of cas-3 and cas-9, Increased Bax and decreased Bcl-2, Induces mitochondrial apoptosis pathway. | 22 |
| HepG2 | 7.5 µM of PAT for 10 h | PAT induces apoptosis, primarily through caspase activation. | 23 |
| HepG2 | 0-80 µM of PAT for 24 h | PAT induces autophagy via the ROS-Akt1-MTOR pathway. | 24 |
| HepG2 | 0-8 µM of PAT for 24 h | PAT induces pyroptosis through the autophagy-cathepsin B-inflammasome pathway. | 25 |
| HepG2 | 2 µM of PAT for 3, 6, 12, or 24 h | PAT induces autophagic cell death through a ROS-Akt1-MTOR pathway. | 26 |
| RAW264.7 | 1 µM of PAT for 8 h | PAT induces mitophagy through p62 and inhibits the activation of innate immune responses. | 27 |
Wu et al. found that PAT induces apoptosis through the intrinsic pathway involving the Bcl-2 family 22. The present findings align with those of Wu and Liu 23, who demonstrated that PAT causes apoptosis in HEK293 and HL-60 cell lines, similar to effects observed with other mycotoxins. Our data indicate that H2452 cancer cells are significantly more sensitive to PAT than MeT-5A healthy cells, as reflected by their IC values. While apoptosis- and autophagy-related genes are modulated in both cell types, cytotoxic and pro-apoptotic effects are more pronounced in cancer cells. PAT is proposed to induce apoptosis by triggering the production of reactive oxygen species (ROS) in many cell lines; for instance, it induces apoptosis in HL-60 and HEK293 cells 9. It has also been demonstrated that PAT can significantly reduce tumor progression in human lung and liver cancer cell lines 7,20.
In the present study, the IC values of PAT were 12.68 µM for H2452 mesothelioma cells and 101.5 µM for MeT-5A healthy mesothelial cells after 24 h of incubation. By comparison, our previous work reported IC values of 8.5 µM for HepG2 liver cancer cells and 10.2 µM for A549 lung cancer cells 24. These data suggest that H2452 cells exhibit intermediate sensitivity to PAT relative to other cancer cell types, whereas healthy mesothelial cells are considerably more resistant. Collectively, these observations underscore the differential cytotoxic effects of PAT across cell types and provide a basis for its selective antitumor activity.
In our previous study, PAT exerted anticancer activity in colon and breast cancer cells at high concentrations, and in neuroblastoma cell lines at both low and high concentrations. Thus, PAT may represent a dose-dependent therapeutic candidate for neuroblastoma, colon, and breast malignancies 25. In HepG-2 hepatocellular carcinoma cells, PAT reduced viability in a dose- and time-dependent manner, from 92 % to 13 % after 24 h and to 2 % after 48 and 72 h of exposure 26. In CHO-K1 cells, proliferation declined from 45 % to 16 % after 24, 48, and 72 h at 3.125 or 6.25 µM 27. A separate CHO-K1 study confirmed a dose-dependent loss of viability and a significant rise in ROS, with IC values of 4.4 µM (NRU) and 0.69 µM (MTS) after 24 h 28. Exposure of HEK293 cells to 2.5–15 µM PAT produced dose-dependent viability reductions of 8 %, 18 %, 31 %, 42 %, and 63 %, respectively 29. PAT (5–100 µM) again decreased viability and concomitantly enhanced ROS generation and p53 expression 30. In H9c2 cardiomyoblasts, the IC was 25 µM, and PAT elevated LDH release and apoptosis 31. Ten-hour treatment of HepG-2 cells with 2.5–15 µM PAT lowered viability from 91.46 % to 53.16 %, with an IC of 15.85 µM 32. In Caco-2 cells, concentrations ≥ 25 µM induced a dose-dependent viability loss (IC = 15.95 µM) 33. Another HepG-2 study reported an IC of 7.2 mM and demonstrated, using modulators of autophagosome formation and ROS, that cytotoxicity was mediated by autophagy 34. Finally, in HCT116 colorectal cancer cells, PAT decreased viability by triggering apoptosis through G/M cell-cycle arrest 35.
Mesothelioma is an aggressive malignancy strongly linked to asbestos exposure and arises predominantly from the mesothelial lining of the pleura, peritoneum, and pericardium 11. The genes SQSTM1 (p62), Beclin-1, ATG5, ATG7, Bax, and Bcl-2 regulate autophagy and apoptosis—fundamental processes that preserve cellular homeostasis under physiological and stress conditions. Deregulation of these pathways can drive oncogenesis, including the development of mesothelioma. p62 serves as a selective autophagy receptor that recognizes ubiquitinated protein aggregates and damaged organelles and targets them for lysosomal degradation. Aberrant expression of these autophagy-related genes has been implicated in cancer, neurodegeneration, and aging. In tumors, impaired autophagic flux allows p62 to accumulate, thereby enhancing oxidative stress and pro-inflammatory signaling 36. In mesothelioma, elevated p62 levels correlate with poor prognosis and are thought to facilitate tumorigenesis by fostering proteotoxic stress and activating inflammatory pathways. Asbestos-induced cellular stress can further up-regulate p62 expression, leading to dysregulated signaling cascades that promote tumor progression 37.
Beclin-1 is a key regulator of autophagy and plays a role in the initiation of autophagosome formation. Decreased Beclin-1 expression has been associated with a defective autophagy process that may promote cancer development by allowing damaged cells to survive and proliferate. Studies have shown that Beclin-1-mediated autophagy plays a protective role in preventing cancer onset. In mesothelioma, decreased Beclin-1 expression may impair autophagic responses and promote tumor progression by predisposing to the accumulation of damaged proteins and organelles 38. Atg5 and Atg7 are required for the formation of autophagosomes and autophagic cell death. → Autophagosome formation and autophagy-related cell death also depend on the ubiquitin-like conjugation enzymes Atg5 and Atg7.
In cancer cells, autophagy can be upregulated to promote cell survival under stress such as nutrient deficiency or hypoxia, which are common in the tumor microenvironment. In mesothelioma, altered autophagic processes due to dysfunctional Atg5 or Atg7 may help cancer cells escape cell death, thereby promoting therapy resistance and tumor progression 39.
Bax is a pro-apoptotic protein, whereas Bcl-2 functions as an anti-apoptotic factor. The ratio between these two proteins determines whether a cell undergoes apoptosis in response to cellular stress or damage. In malignant mesothelioma, deregulated Bax/Bcl-2 expression can impair apoptosis, allowing tumor cells to survive despite lethal stimuli. Overexpression of Bcl-2 and/or down-regulation of Bax has been linked to chemoresistance and radioresistance in mesothelioma 40.
Dysregulated autophagy is also documented in mesothelioma. Crosstalk between autophagy and apoptosis is complex; alterations in Beclin-1, Atg5, Atg7, or p62/SQSTM1 disrupt autophagic flux and favour tumorigenesis 39. Concomitant defects in apoptotic signalling caused by an imbalanced Bax/Bcl-2 axis further enhance cell survival and contribute to therapeutic resistance. Consequently, expression and interaction of these genes are critical determinants of tumour initiation, progression, and response to therapy. Elucidating their roles may identify new therapeutic targets aimed at restoring physiological autophagy and apoptosis in mesothelioma cells.
In mesothelioma cells, Patulin (PAT) treatment reduced Bax and Bcl-2 expression. Paradoxically, decreased Bax may lower the propensity for apoptosis, thereby promoting survival, whereas reduction of Bcl-2—typically overexpressed in cancers—may represent a failed compensatory attempt to relieve apoptotic blockade; nonetheless, the net effect remains suppression of caspase-mediated cell death 41.
PAT exposure concomitantly increased Atg5 and Atg7 transcript levels, suggesting a shift toward an autophagy-dependent death pathway. However, reduced Beclin-1 and SQSTM1/p62 expression indicates that, although autophagy initiation is triggered, downstream execution is defective, potentially compromising the adaptive response to PAT-induced stress 42.
In non-malignant mesothelial cells, as in their malignant counterparts, exposure to PAT resulted in reduced Bax and Bcl-2 expression. This concurrent down-regulation likely reflects interference with the apoptotic machinery that neither fully commits the cells to apoptosis nor preserves them but instead diverts them toward alternative death or survival programs. In MeT-5A cells, the simultaneous decrease in Beclin-1, SQSTM1, and Atg7 indicates impaired autophagy, a pattern not observed in H2452 cells. Diminished Beclin-1 and Atg7 may impede the initiation and elongation of the autophagic cascade, thereby favoring cellular damage rather than cytoprotection. Conversely, Atg5 was up-regulated in MeT-5A cells, suggesting partial, non-productive activation of autophagy.
Collectively, PAT modulates apoptotic and autophagic signalling in both normal and malignant mesothelial cells. In tumor cells, this modulation appears to support survival by attenuating apoptosis and enhancing components of the autophagic machinery, potentially enabling mesothelioma cells to withstand stress. In MeT-5A cells, however, PAT simultaneously suppresses key apoptotic and autophagic genes—particularly Beclin-1, Atg7, and SQSTM1—thereby disturbing cellular homeostasis; the isolated rise in Atg5 may represent an abortive autophagic attempt.
Overall, PAT treatment alters the transcription of critical autophagy- and apoptosis-related genes (Bax, Bcl-2, Beclin-1, SQSTM1, Atg5, and Atg7). Up-regulation of Atg5 and Atg7, accompanied by down-regulation of Beclin-1 and SQSTM1, denotes a nuanced reprogramming of the autophagy network. Such gene-expression changes probably signify an adaptive stress response rather than unequivocal activation or suppression of autophagy or apoptosis.
Although we observed significant changes in the mRNA levels of autophagy-related genes after PAT treatment, protein-level validations and functional autophagy flux assays were not conducted in this study. Therefore, additional investigations are necessary to confirm these transcriptional findings at the protein and functional levels.
Conclusion
The mesothelium is essential for the embryonic development of organs such as the heart, lungs, and intestines within the coelomic cavity. A range of pathological conditions can compromise mesothelial derivatives, the most severe being malignant mesothelioma. Genes implicated in mesothelial differentiation and homeostasis act as diagnostic biomarkers for mesothelioma, facilitating its distinction from other thoracic malignancies. Despite asbestos bans in numerous countries, the incidence of malignant pleural mesothelioma (MPM) remains persistent. Ongoing asbestos exposure represents the principal etiological factor driving MPM. Therapeutic development for mesothelioma has lagged behind that for other solid tumours, leaving patients with few effective options. Although immune-checkpoint and anti-angiogenic strategies yield promising results, an urgent demand for novel, efficacious modalities persists. PAT exposure modulated the transcription of apoptosis- and autophagy-associated genes in H2452 and MeT-5A cells, suggesting interference at critical regulatory nodes. However, corroboration at the protein level and assessment of autophagic flux are required to validate these transcriptional findings. Collectively, these observations underscore the complex regulatory actions of PAT and justify additional mechanistic studies.
Abbreviations
ATG5: Autophagy related 5; ATG7: Autophagy related 7; Bax: BCL2 associated X protein; Bcl-2: B-cell lymphoma 2; BECN-1: Beclin-1; cDNA: Complementary Deoxyribonucleic Acid; DMEM: Dulbecco's Modified Eagle Medium; DMSO: Dimethyl Sulfoxide; FBS: Fetal Bovine Serum; IARC: International Agency for Research on Cancer; IC: Half Maximal Inhibitory Concentration; LDH: Lactate Dehydrogenase; MM: Malignant Mesothelioma; MPM: Malignant Pleural Mesothelioma; MTS: 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium; PAT: Patulin; PCR: Polymerase Chain Reaction; ROS: Reactive Oxygen Species; RQ: Relative Quantification; RT-PCR: Reverse Transcription Polymerase Chain Reaction; SQSTM1: Sequestosome 1; WHO: World Health Organization
Acknowledgments
Prof. Dr. Asuman Demiroğlu-Zergeroğlu's help in providing the Met 5A cell line and Prof. Dr. Sabahattin Cömertpay's help in providing the H2452 cell line are gratefully acknowledged.
Author’s contributions
The research was conceptualized and designed by Songül Ünüvar. Rümeysa Akkutlu, Hande Yüce, Dilan A. Özek, and Yasemin Berberoğlu carried out the experiments. Hande Yüce, Dilan A. Özek, and Yasemin Berberoğlu examined the information. Songül Ünüvar wrote the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported by TUBITAK 2209-A (The Scientific and Technological Research Council of Türkiye, Project No. 2209-A, Project ID: 1919B012203014).
Availability of data and materials
Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Declaration of generative AI and AI-assisted technologies in the writing process
The authors declare that they have not used generative AI (a type of artificial intelligence technology that can produce various types of content including text, imagery, audio and synthetic data. Examples include ChatGPT, NovelAI, Jasper AI, Rytr AI, DALL-E, etc) and AI-assisted technologies in the writing process before submission.
Competing interests
The authors declare that they have no competing interests.

