Evolving Genetic Research in Rheumatoid Arthritis: Mapping Research Trends and Global Contributions
- Human Genome Centre, School of Medical Sciences, Health Campus
- Hospital Pakar Universiti Sains Malaysia, 16150 Kubang Kerian, Kelantan, Malaysia
- Department of Internal Medicine, School of Medical Sciences, Health Campus
- Department of Immunology, School of Medical Sciences, Health Campus
- Department of Health Information Management, Universitas Indonesia Maju, Jakarta, Indonesia
Abstract
Background: Significant advancements in genetic research have profoundly expanded our understanding of rheumatoid arthritis, elucidating numerous loci and pathways associated with disease susceptibility, progression, and therapeutic response. Methods: This bibliometric study mapped global research trends in genetics and rheumatoid arthritis using 3,731 publications indexed in Scopus from 1953 to June 17, 2025. Publications from 2025 were excluded from the total publication and citation trend analyses owing to incomplete data. Results: Publication volume ascended progressively from the 1990s, peaking in 2022. China leads in total output, while the United States demonstrates the highest citation impact and research influence. The United States, the United Kingdom, the Netherlands, Sweden, and Spain emerged as pivotal research hubs, contributing the majority of the most prolific authors and highly cited articles. Key publishing platforms include Arthritis and Rheumatism and Annals of the Rheumatic Diseases. Keyword co-occurrence analysis revealed dynamic shifts in research priorities: traditional topics such as Human Leukocyte Antigen (HLA), shared epitope, and rheumatoid factors are waning, while emerging domains include machine learning, ribonucleic acid sequencing (RNA-seq), Mendelian randomization, genome-wide association studies (GWAS), bioinformatics, and biomarkers. Conclusion: These findings highlight the evolving paradigm of rheumatoid arthritis genetics research and offer insights to guide future investigations and bolster international collaboration.
INTRODUCTION
Rheumatoid arthritis is a systemic, chronic inflammatory pathology and an autoimmune disorder1. It is characterized by progressive and destructive arthropathy stemming from synovial inflammation and hyperplasia, along with an elevation in autoantibodies such as rheumatoid factor (RF) and anti-cyclic citrullinated peptide (anti-CCP)2. In addition to musculoskeletal involvement, rheumatoid arthritis is associated with extra-articular manifestations, particularly cardiovascular disease, which contributes to diminished life expectancy among affected individuals3. Rheumatoid arthritis is a complex polygenic disease influenced by the interplay of genetic predisposition and environmental triggers, both of which play instrumental roles in disease susceptibility and progression4.
Significant strides in genetic research have markedly enhanced our understanding of rheumatoid arthritis pathogenesis, identifying numerous loci and molecular pathways involved in disease susceptibility, severity, and therapeutic response5. These insights have highlighted critical immune-related cell types and mechanisms, providing the groundwork for personalized medicine approaches. Current and future challenges include leveraging genetic variants as instrumental variables to establish causal relationships with intermediate phenotypes (e.g., transcriptomic markers), developing polygenic risk scores for diagnosis and treatment stratification, and identifying novel therapeutic targets4.
While interest in rheumatoid arthritis genetics continues to intensify, bibliometric analyses of this expanding research domain remain limited. Extant studies have primarily focused on specific subtopics, such as deoxyribonucleic acid (DNA) methylation6, non-coding ribonucleic acids (RNA)7, genome-wide association studies (GWAS)8, RNA-binding proteins9, and microRNAs10. However, a comprehensive evaluation of the global research landscape concerning rheumatoid arthritis genetics is still lacking. This study aims to fill that gap by mapping publication trends, research impact, collaborative networks, leading journals, influential authors, the most frequently cited articles, and the thematic evolution within the field. By offering an in-depth bibliometric overview, this analysis provides valuable insights that can guide future research directions and inform clinical applications11,12.
METHODS
Database selection
Scopus was selected as the primary data source for this study due to its comprehensive coverage of global scientific output. Widely recognized by the international research community, Scopus is considered a reliable, high-quality bibliometric database for conducting quantitative analyses in academic research13,14.
Search strategy
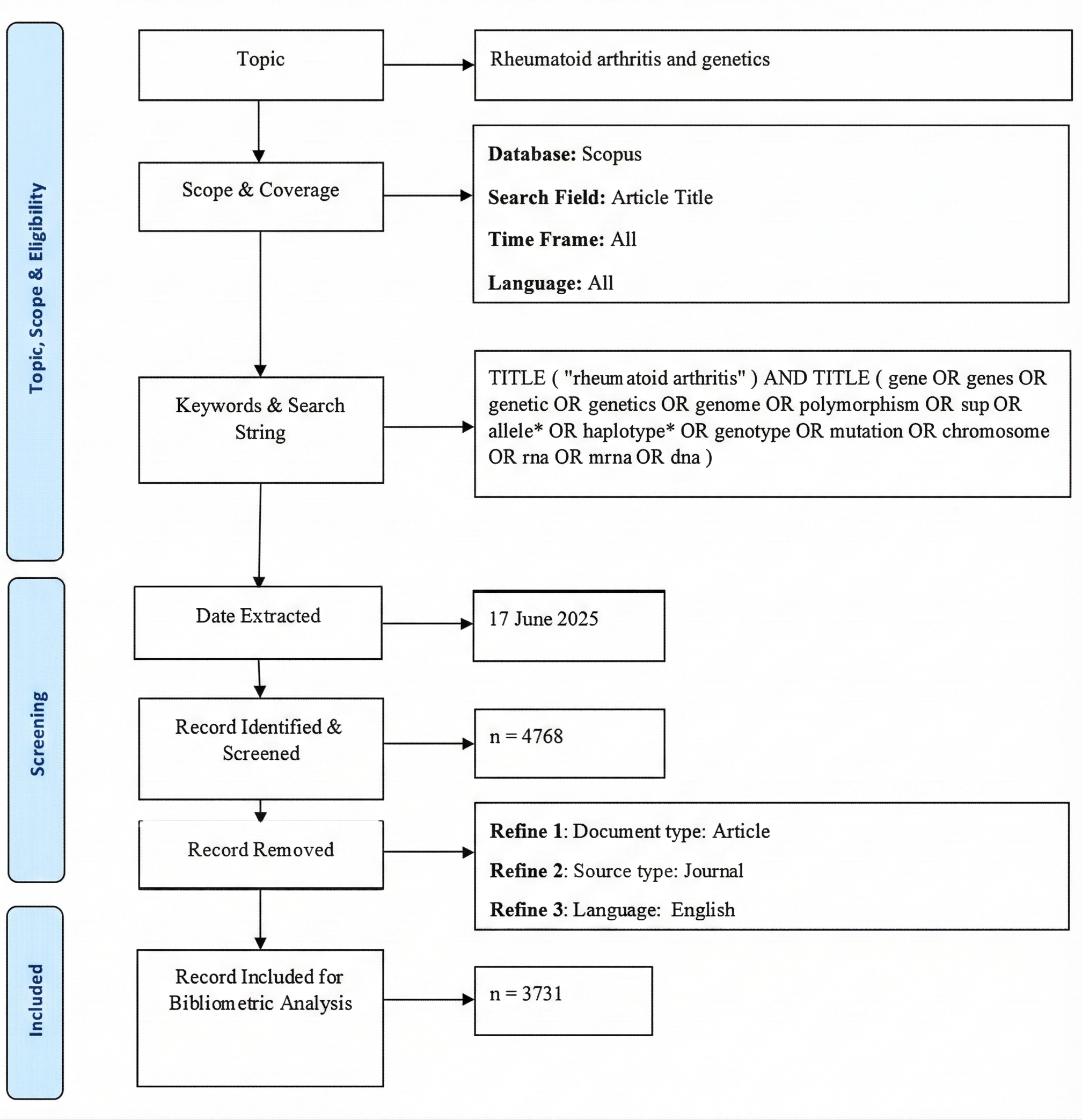
A systematic search was conducted in Scopus on June 17, 2025, using the query: TITLE ("rheumatoid arthritis") AND TITLE (gene OR genes OR genetic OR genetics OR genome OR polymorphism OR snp OR allele* OR haplotype* OR genotype OR mutation OR chromosome OR rna OR mrna OR dna). The TITLE field was utilized in the Scopus search to enhance specificity by retrieving only articles explicitly focused on the genetic aspects of rheumatoid arthritis. This minimized the inclusion of studies where genetic terms appear incidentally in abstracts or keywords, thereby reducing noise and sharpening the analytical focus. The search strategy was validated by reviewing the relevance of the retrieved articles. Only English-language original research articles focusing on the genetic aspects of rheumatoid arthritis were included, while reviews, conference papers, editorials, letters, and non-English publications were excluded. A total of 3,731 documents represent the eligible records retrieved directly from Scopus after filtering. Figure 1 depicts the PRISMA flow diagram of the study screening and selection process.
Flow diagram of search strategy
Data Analysis
Citation analysis was performed using Harzing’s Publish or Perish software (version 8)16 on data exported in RIS format, from which citation metrics were extracted. Country co-authorship and author keywords co-occurrence maps were generated using VOSviewer (version 1.6.20)17 based on data in CSV format. For all VOSviewer analyses, the default resolution parameter (resolution = 1.0) was applied. In the country co-authorship analysis, a minimum threshold of five documents per country was set, resulting in the inclusion of 62 countries in the network visualization. For the author keyword co-occurrence analysis18, a minimum occurrence threshold of 10 occurrences per keyword was applied. To improve analytical accuracy and reduce term fragmentation, a thesaurus file was employed to standardize synonymous and variant terms, including singular and plural forms as well as commonly used abbreviations. For example, “cytokine” was merged with “cytokines,” “major histocompatibility complex” with “MHC,” and “interleukin-1” with “IL-1”. This standardization enhanced the precision and interpretability of the VOSviewer analyses.
RESULTS
A total of 3,731 documents were included in the final analysis to map publication trends, research impact, collaborative networks, leading journals, influential authors, the most cited articles, and the thematic evolution within the field.
Annual Trends in Scientific Publications and Citations
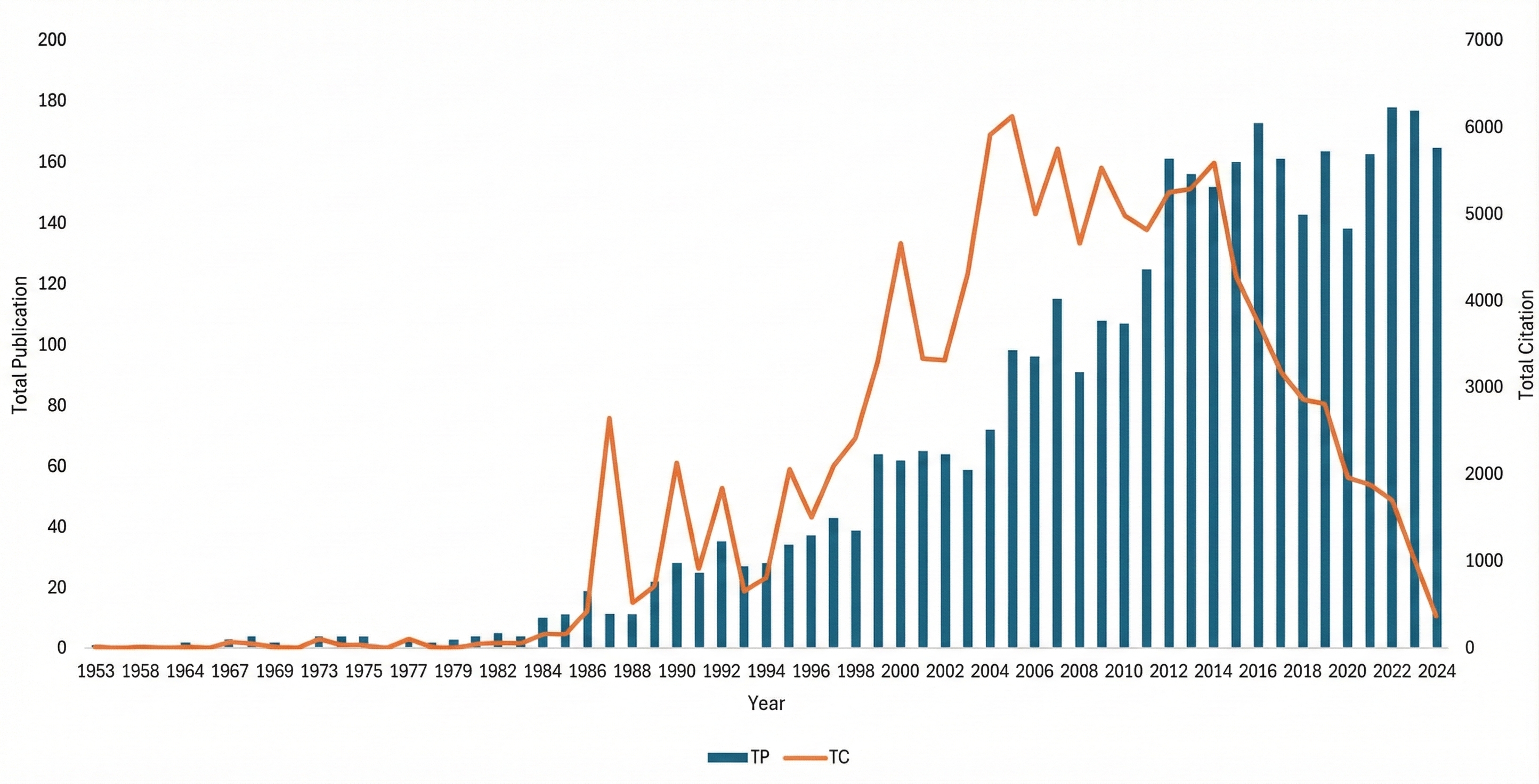
The annual trends in scientific publications and citations from 1953 to 2024 demonstrate a clear evolution in research activity (Figure 2). Early output was negligible, with fewer than five papers per year until the 1980s. A gradual rise began in the late 1980s, marked by a spike in 1987, when 11 publications received 2,659 citations, reflecting the influence of seminal works. A sustained increase followed through the 1990s, highlighting growing interest in the field. The most productive epoch occurred between 2000 and 2015, with publication volume and citation counts peaking at 173 papers in 2016 and 6,146 citations in 2005. Although annual output remained high beyond 2016, citations have declined slightly, likely due to the citation lag for newer publications. Despite this, the continued high publication rate suggests enduring interest in this research area.
Trends in the number of publications and citations from 1953 to 2024 (year 2025 is not shown because of incomplete data)
Country Contributions to Publications and International Collaboration Analysis
Table 1 presents the top 10 contributing countries in rheumatoid arthritis and genetics research based on bibliometric indicators. China leads in total publications (663), followed by the United States (608) and the United Kingdom (409). Despite producing fewer papers than China, the United States ranks highest in citation impact, with the most total citations (38,878), the highest average citations per publication (63.94), and the top h-index (95), reflecting both the volume and influence of its research output. European countries, such as Sweden, the Netherlands, and the United Kingdom, demonstrate strong citation performance relative to their output. Sweden, in particular, attained the highest average citations per publication (87.31) and cited publication (90.41), indicating high-impact research despite fewer publications. Meanwhile, East Asian countries like Japan, South Korea, and China contribute substantially to publication volume, though citation rates vary—Japan shows moderate impact (45.03), while China’s lower average (17.65) suggests a growing but still maturing influence.
Top 10 countries that contributed to the publications
| Country | TP | NCP | TC | C/P | C/CP | ||
|---|---|---|---|---|---|---|---|
| China | 663 | 594 | 11703 | 17.65 | 19.70 | 43 | 78 |
| United States | 608 | 587 | 38878 | 63.94 | 66.23 | 95 | 172 |
| United Kingdom | 409 | 392 | 25240 | 61.71 | 64.39 | 77 | 142 |
| Japan | 263 | 250 | 11844 | 45.03 | 47.38 | 55 | 100 |
| Spain | 238 | 230 | 10520 | 44.20 | 45.74 | 48 | 91 |
| Netherlands | 213 | 208 | 16967 | 79.66 | 81.57 | 65 | 125 |
| France | 183 | 175 | 9037 | 49.38 | 51.64 | 44 | 90 |
| Sweden | 175 | 169 | 15279 | 87.31 | 90.41 | 58 | 121 |
| South Korea | 168 | 164 | 7330 | 43.63 | 44.70 | 40 | 79 |
VOSviewer was utilized to visualize the international co-authorship network. As shown in Figure 3, the analysis encompassed 62 countries with at least five publications, stratified into seven clusters based on co-authorship frequency. These clusters underscore distinct regional and thematic research collaborations, with each cluster representing a group of countries more likely to publish together. The United States (turquoise cluster), along with the United Kingdom, Netherlands, and Sweden (red cluster), and Spain (turquoise cluster), stood out as key contributors, showing both high publication output and strong citation impact. Their high total link strength values suggest central roles in global research networks. In contrast, countries like Thailand and Lithuania show weaker international collaboration, highlighting room for growth. Emerging nations such as China, Iran, Pakistan, and several Middle Eastern countries (yellow nodes) are becoming more active, with increasing publication counts. Although many of their outputs are recent, resulting in lower average citations, their growing publication volume suggests strong potential for future impact.
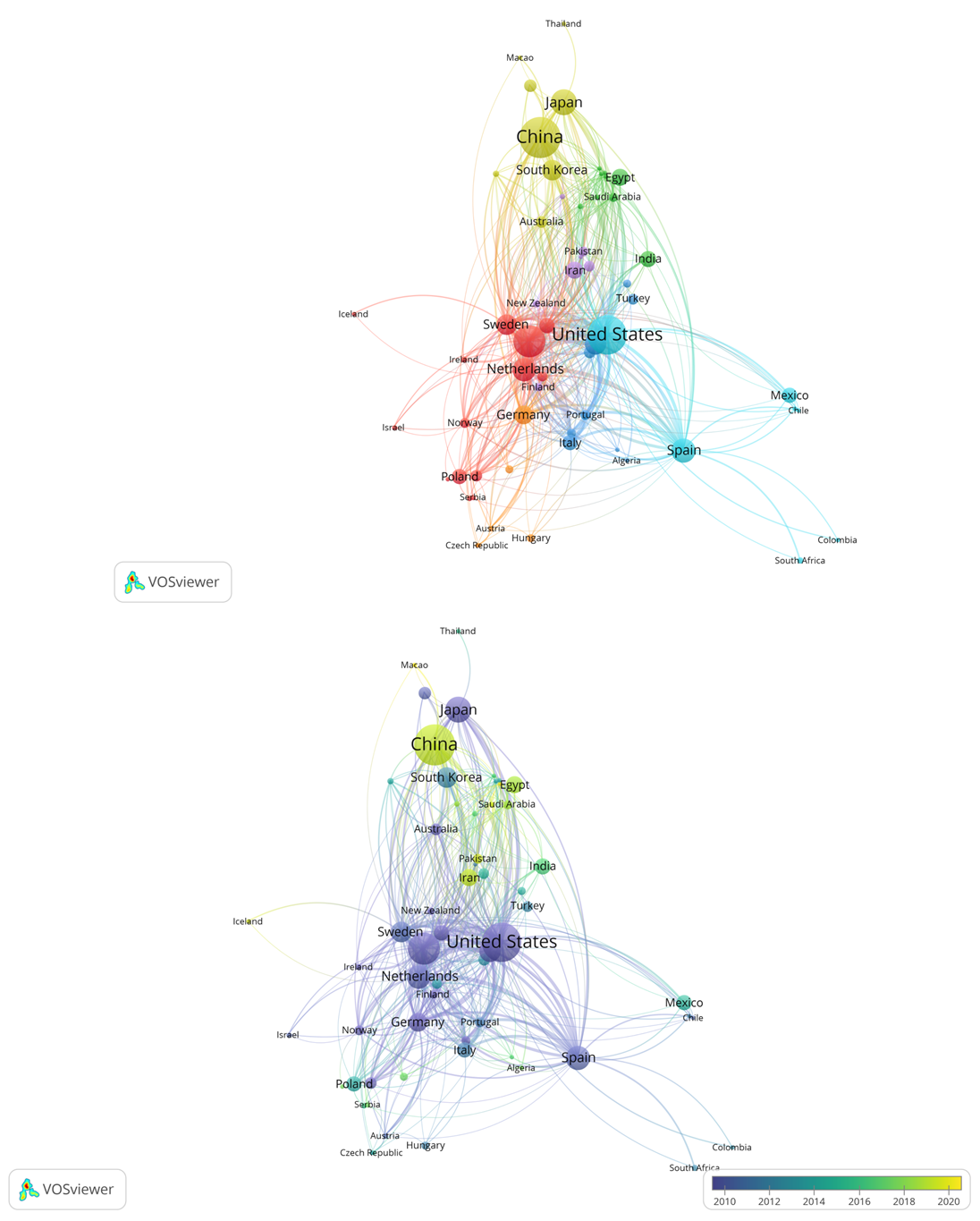
Co-authorship map of countries in rheumatoid arthritis and genetics research. (a) Network visualization showing collaboration clusters among countries; node size represents the number of documents, and link thickness indicates the strength of co-authorship relationships. (b) Overlay visualization illustrating the temporal evolution of country collaborations, where node size reflects publication output and node color (purple to yellow) represents the average publication year (2005–2021)
Leading Journals
Table 2 highlights the top ten journals contributing to rheumatoid arthritis and genetics research, emphasizing both publication volume and citation impact. Arthritis and Rheumatism leads in output with 302 articles and over 25,000 citations, reflecting its historical significance, though it has since transitioned to Arthritis and Rheumatology. Its legacy is underscored by strong metrics, including a CiteScore of 21.4. In current influence, Annals of the Rheumatic Diseases stands out with the highest CiteScore (33.2) and a robust citation profile, ranking second in volume but maintaining top-tier impact. Other key contributors include Journal of Rheumatology, Arthritis Research and Therapy, and Rheumatology, each showing strong bibliometric performance. Arthritis Research and Therapy, for example, garnered over 5,000 citations from 131 papers, supported by a CiteScore of 8.5. Generalist and interdisciplinary journals like PLOS One and Frontiers in Immunology also feature prominently, reflecting the expanding interface between rheumatology, immunology, and broader biomedical research. Frontiers in Immunology, despite fewer papers, demonstrates high impact with a CiteScore of 10.8 and strong visibility across disciplines.
Top 10 Most Productive Journals
| Source Title | TP | TC | Publisher | Cite Score (2024) | SJR (2024) | SNIP (2024) |
|---|---|---|---|---|---|---|
| Arthritis and Rheumatism* | 302 | 25908 | John Wiley & Sons | 21.4* | 3.784* | 2.788* |
| Annals of the Rheumatic Diseases | 190 | 9466 | Elsevier | 33.2 | 5.731 | 4.435 |
| Journal of Rheumatology | 167 | 4929 | Journal of Rheumatology | 5.9 | 1.135 | 1.028 |
| Arthritis Research and Therapy | 131 | 5010 | Springer Nature | 8.5 | 1.587 | 1.274 |
| Rheumatology | 105 | 3798 | Oxford University Press | 9.9 | 1.721 | 1.373 |
| Rheumatology International | 105 | 2060 | Springer Nature | 7.0 | 0.965 | 1.155 |
| Clinical and Experimental Rheumatology | 99 | 1853 | Clinical and Experimental Rheumatology S.A.S. | 5.9 | 1.004 | 0.952 |
| Plos One | 93 | 2231 | Public Library of Science | 5.4 | 0.803 | 1.065 |
| Clinical Rheumatology | 84 | 1107 | Springer Nature | 5.9 | 0.855 | 0.943 |
| Frontiers in Immunology | 62 | 1059 | Frontiers Media S.A. | 10.8 | 1.941 | 1.259 |
Most Prolific Authors
Table 3 highlights the top ten most prolific authors, showcasing their affiliations, countries, and bibliometric impact. Martín J (Spain) leads in total publications (99) and cited papers (97), reflecting both productivity and consistent scholarly influence. Barton A (UK), despite fewer publications (73), achieves the highest average citations per paper (110.90), emphasizing the impact of his contributions. Other highly influential figures include Gregersen PK (USA) with the most total citations (12,849), and Huizinga TWJ (Netherlands), with a remarkable average citations per paper of 132.62. Swedish researchers Klareskog L and Padyukov L stand out with high citation averages (155.00 and 163.22, respectively), reinforcing Sweden's research prominence. Spanish authors like Balsa A and González-Gay MA further underscore Spain’s active role in the field, while Bae SC and Lee YH represent South Korea’s growing influence through consistent output and strong citation metrics.
Top 10 Most Prolific Authors
| Author’s Name | Affiliation | Country | TP | NCP | TC | C/P | C/CP | ||
|---|---|---|---|---|---|---|---|---|---|
| Martín J | CSIC - Instituto de Parasitologia y Biomedicina Lopez Neyra (IPBLN) | Spain | 99 | 97 | 6056 | 61.17 | 62.43 | 37 | 76 |
| Balsa A | Hospital Universitario La Paz | Spain | 83 | 83 | 3295 | 39.70 | 39.70 | 31 | 54 |
| Bae SC | Hanyang University | South Korea | 82 | 82 | 4573 | 55.77 | 55.77 | 29 | 66 |
| Barton A | University of Manchester | United Kingdom | 73 | 71 | 8096 | 110.90 | 114.03 | 41 | 73 |
| González-Gay MA | Universidad de Cantabria | Spain | 71 | 70 | 4643 | 65.39 | 66.33 | 27 | 68 |
| Klareskog L | Karolinska Institutet | Sweden | 69 | 66 | 10695 | 155.00 | 162.05 | 42 | 69 |
| Lee YH | Korea University Medicine | South Korea | 69 | 68 | 2104 | 30.49 | 30.94 | 25 | 43 |
| Gregersen PK | Northwell Health System | United States | 65 | 65 | 12849 | 197.68 | 197.68 | 38 | 65 |
| Huizinga TWJ | Leids Universitair Medisch Centrum | Netherlands | 65 | 64 | 8620 | 132.62 | 134.69 | 41 | 65 |
| Padyukov L | Karolinska Universitetssjukhuset | Sweden | 60 | 59 | 9793 | 163.22 | 165.98 | 35 | 60 |
Most Influential Articles
Table 4 highlights the 10 most highly cited publications in rheumatoid arthritis and genetics, representing foundational work that has shaped current scientific understanding. Leading the list is Gregersen et al. (1987)19, and their shared epitope hypothesis remains a seminal contribution with 2,265 citations and a lasting impact on molecular rheumatoid arthritis research. Okada et al. (2014)20, published in Nature, with 1,807 citations and the highest annual citation rate (164.27), marking a transformative step in linking GWAS findings to therapeutic development. Similarly, Begovich et al. (2004)21 identified a key single-nucleotide polymorphism (SNP) in protein tyrosine phosphatase non-receptor type 22 (PTPN22), earning 1,256 citations and influencing subsequent genetic studies on autoimmune susceptibility. Several other top-cited articles delve into GWAS and functional genomics, such as those by Stahl et al. (2010)22 and Suzuki et al. (2003)23, which expanded the map of rheumatoid arthritis-associated loci. Padyukov et al. (2004)27 made a pivotal contribution by linking smoking with Human Leukocyte Antigen (HLA)–DR alleles, offering early evidence of gene–environment interactions. Liu et al. (2013)25 brought epigenetics to the forefront with insights into DNA methylation in rheumatoid arthritis. Earlier landmark studies by MacGregor et al. (2000)24 on heritability and Firestein et al. (1990)26 on cytokine gene expression continue to be influential, reflecting their foundational role in both clinical and molecular aspects of rheumatoid arthritis research.
Top 10 influential articles
| Authors and year | Title | Cites | Cites/Year |
|---|---|---|---|
| Gregersen | The shared epitope hypothesis. an approach to understanding the molecular genetics of susceptibility to rheumatoid arthritis. | 2265 | 59.58 |
| Okada | Genetics of rheumatoid arthritis contributes to biology and drug discovery. | 1807 | 164.27 |
| Begovich | A missense single-nucleotide polymorphism in a gene encoding a protein tyrosine phosphatase (PTPN22) is associated with rheumatoid arthritis. | 1256 | 59.81 |
| Stahl | Genome-wide association study meta-analysis identifies seven new rheumatoid arthritis risk loci. | 1087 | 72.47 |
| Suzuki | Functional haplotypes of PADI4, encoding citrullinating enzyme peptidylarginine deiminase 4, are associated with rheumatoid arthritis. | 978 | 44.45 |
| MacGregor | Characterizing the quantitative genetic contribution to rheumatoid arthritis using data from twins. | 956 | 38.24 |
| Liu | Epigenome-wide association data implicate DNA methylation as an intermediary of genetic risk in rheumatoid arthritis. | 779 | 64.92 |
| Firestein | Quantitative analysis of cytokine gene expression in rheumatoid arthritis. | 567 | 16.20 |
| Padyukov | A gene–environment interaction between smoking and shared epitope genes in HLA–DR provides a high risk of seropositive rheumatoid arthritis. | 560 | 26.67 |
| Tokuhiro | An intronic SNP in a RUNX1 binding site of SLC22A4, encoding an organic cation transporter, is associated with rheumatoid arthritis. | 539 | 24.50 |
Visualization Analysis of Dynamic Changes in Co-Occurrence of the Author’s Keywords
VOSviewer was used to generate a keyword co-occurrence map based on 102 author keywords grouped into four thematic clusters (Figure 4). Cluster 1 (red) is the largest and most recent cluster, focusing on the molecular underpinnings of rheumatoid arthritis and the development of personalized treatment strategies. Prominent keywords include rheumatoid arthritis, methotrexate, inflammation, autoimmune diseases, gene expression, biomarkers, and cytokines. Emerging keywords like bioinformatics (2021.8), machine learning (2023.1), and RNA-seq (2020.1) reflect a data-driven shift in rheumatoid arthritis research. Recent interests include Mendelian randomization (2023.3), COVID-19 (2022.7), and interstitial lung disease (2022.9). The green cluster (Cluster 2) captures classical genetic epidemiology and comparative autoimmune research. Recurring keywords are polymorphisms, SNPs, meta-analysis, systemic lupus erythematosus (SLE), and autoantibodies. Although many terms are older (e.g., juvenile rheumatoid arthritis, 2002), their continued relevance reflects enduring efforts in gene discovery. The blue cluster (Cluster 3) represents well-established immunogenetic domains with clinical relevance. Main keywords include HLA, HLA-DRB1, rheumatoid factors, anti-CCP, PTPN22, and shared epitope. Keywords such as cardiovascular disease, atherosclerosis, and disease activity reflect links between genetics and clinical outcomes. Many keywords are either older (e.g., HLA, 2004.2) or more recent, such as anti-CCP (2016.0) and ACPA (2016.2). The yellow cluster (Cluster 4) explores functional polymorphisms and genotype-phenotype relationships. The keywords include gene polymorphisms, haplotypes, genotypes, tumor necrosis factor (TNF), vitamin D receptor (VDR), etanercept, and TNF-α. Some keywords, such as vitamin D (2019.4) and TNF-α (2018.5), reflect recent interest in functional gene variants. This cluster also contains older keywords related to foundational methods like restriction fragment length polymorphism (RFLP, 2005.5) and immunogenetics (2002.3).
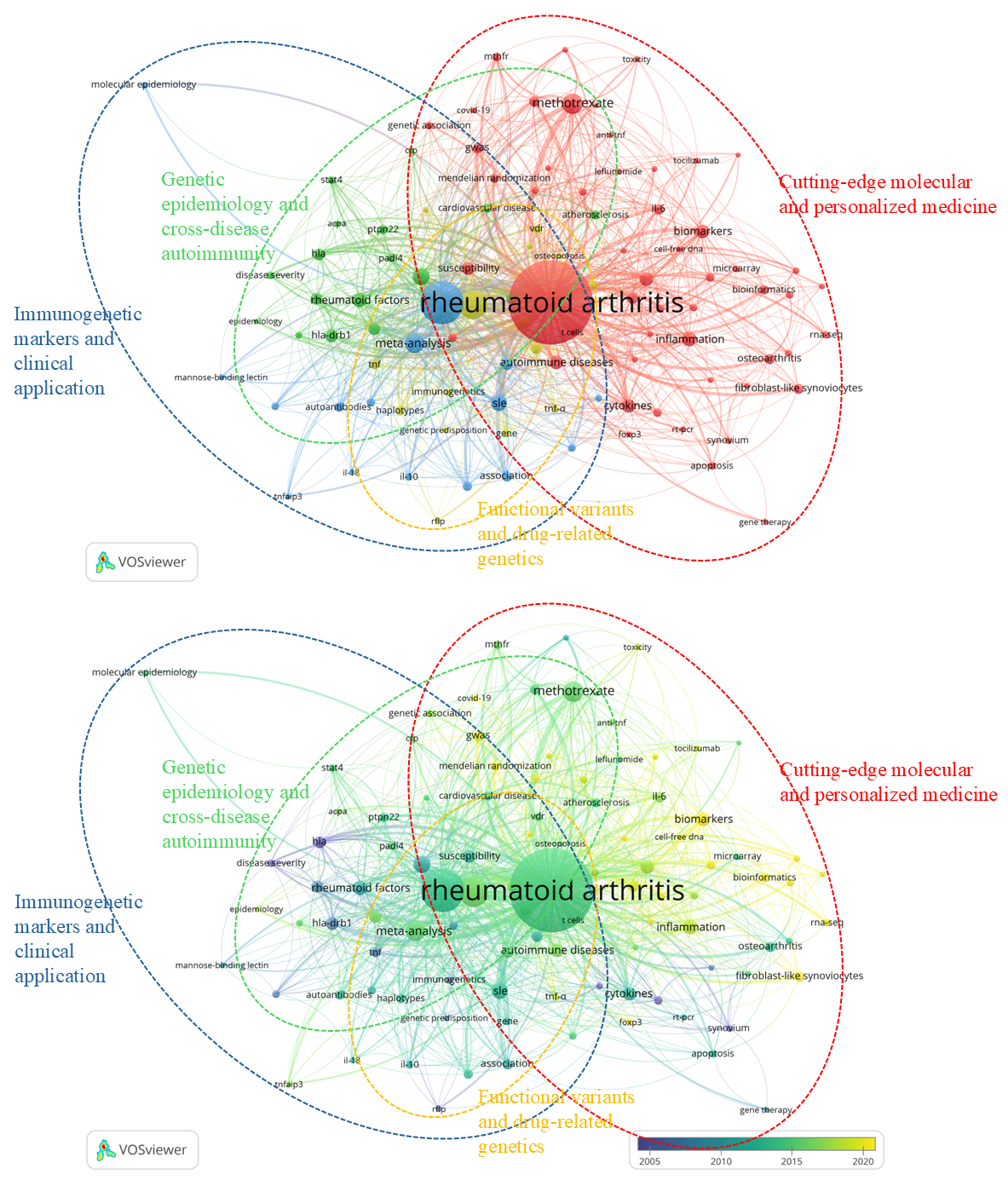
Co-occurrence of author keywords in rheumatoid arthritis and genetics research. (a) Network visualization displaying keyword clusters based on co-occurrence relationships; node size indicates the frequency of keyword occurrence, and link thickness represents the strength of co-occurrence. (b) Overlay visualization showing the temporal distribution of keywords, with node color (purple to yellow) indicating the average publication year
DISCUSSION
Our results indicate a clear trend of growth in the number of publications on rheumatoid arthritis and genetics over time. The annual trends from 1953 to 2024 reveal a steady evolution, beginning with negligible publication rates until the 1980s. A pivotal turning point occurred in 1987, when a high-impact study on the shared epitope hypothesis by Gregersen et al. generated a significant citation spike, signaling foundational contributions to the field. Research activity accelerated throughout the 1990s, culminating in the most productive epoch between 2000 and 2015. This rise may have been facilitated by the concurrent launch of the Human Genome Project (HGP) in October 1990. The HGP was a multinational project involving the United States, the United Kingdom, France, Japan, Canada, Germany, and China, undertaking to sequence the entire human genome and classify human genes 29. Although citations have slightly declined since 2016, likely due to the natural lag in citation accrual, publication rates have remained consistently high, indicating sustained global interest and active investigation in this domain.
The analysis of country contributions and international collaboration highlights both established and emerging global players. While China leads in publication volume, the United States excels in citation impact and research influence, as reflected in its top average citations per publication and h-index. European countries like Sweden and the Netherlands also show high citation efficiency, producing fewer but highly impactful papers. VOSviewer’s co-authorship network visualization indicates that these influential nations are central to global collaboration clusters, with the United States, United Kingdom, Netherlands, Sweden, and Spain forming key hubs. In contrast, countries with lower publication output, such as Thailand and Lithuania, display limited international connectivity. However, emerging contributors, particularly China, Iran, Pakistan, and several Middle Eastern nations, are becoming more active, indicating expanding global participation and growing research potential despite currently modest citation impact. Notably, China’s high publication output is largely driven by large-scale initiatives like the China Kadoorie Biobank (CKB), GWAS consortia, and other cohort studies.
The leading journals, authors, and most-cited articles reveal a tightly interlinked landscape of scholarly influence, scientific advancement, and interdisciplinary growth. High-impact journals like Arthritis and Rheumatism and Annals of the Rheumatic Diseases serve as key publication venues for seminal works, such as Gregersen et al. (1987)19, MacGregor et al. (2000)24, and Padyukov et al. (2004)27, highlighting their role in disseminating groundbreaking genetic discoveries. The prominence of generalist outlets like PLOS One and immunology-focused platforms like Frontiers in Immunology signals the increasing cross-pollination between rheumatology, genomics, and broader biomedical research. This aligns with the evolving scope of top-cited papers, which span from early heritability studies to cutting-edge GWAS, epigenetics, and gene–environment interactions. Importantly, this pattern is not only of academic interest but also highlights the translational trajectory of rheumatoid arthritis genetics research. For instance, GWAS that identified variants in the Janus kinase (JAK)- signal transducer and activator of transcription (STAT) or JAK–STAT signaling pathway provided the scientific foundation for the development of JAK inhibitors such as tofacitinib and baricitinib, which are now approved for clinical use in rheumatoid arthritis5,20,30.
A strong correlation also emerges between the productivity of influential authors and the top-cited articles. Researchers such as Martín (Spain), Gregersen (United States), and Klareskog (Sweden), all of whom appear in the top 10 author rankings, are also behind some of the most cited articles, reinforcing their leadership in shaping the field. The keyword co-occurrence analysis using VOSviewer reveals a rich and evolving thematic structure in rheumatoid arthritis and genetics research, segmented into four distinct yet interconnected clusters. Cluster 1 (red) emerges as the largest and most recent, showcasing the field’s shift toward molecular precision and personalized therapy31,32,33. Core concepts such as rheumatoid arthritis, methotrexate, gene expression, and cytokines anchor this cluster in the disease's biological basis and treatment landscape. However, the appearance of newer keywords like machine learning, RNA-seq, and bioinformatics signals a growing reliance on high-throughput and computational methodologies to dissect complex genetic patterns34,35,36. The inclusion of Mendelian randomization, COVID-19, and interstitial lung disease further reflects an expanding research scope that now incorporates global health concerns and comorbidity profiling37,38,39.
The second largest cluster (green) emphasizes polymorphisms, SNPs, and meta-analysis, as well as comparisons with related autoimmune diseases like SLE40,41. The persistent use of older keywords such as juvenile rheumatoid arthritis underscores how foundational studies in genetic susceptibility continue to inform current hypotheses. These insights highlight opportunities for future studies to explore shared genetic mechanisms across autoimmune conditions and to refine genotype-based risk stratification models42,43. Clinically, such advancements could support the development of more targeted diagnostic tools and personalized therapeutic strategies in rheumatoid arthritis and related disorders44. In contrast, Cluster 3 (blue) focuses on well-established immunogenetic hallmarks that continue to shape clinical practice. Keywords like HLA, shared epitope, and PTPN22 are deeply embedded in rheumatoid arthritis's pathogenic narrative45,46, while the presence of anti-CCP, cardiovascular disease, and disease activity highlights a maturing focus on translational outcomes47,48. Lastly, Cluster 4 (yellow) bridges basic and translational science through exploration of functional gene variants and genotype–phenotype relationships. Keywords like VDR, etanercept, and TNF-α underscore the genetic underpinnings of therapeutic response49,50,51, with vitamin D and gene polymorphisms suggesting newer angles on immune modulation52,53. The coexistence of older methods such as RFLP and immunogenetics with newer molecular targets illustrates the ongoing synthesis of traditional genetic tools with modern approaches54,55.
Beyond bibliometric trends, a closer look at the four keyword clusters reveals both consensus and ongoing controversies in rheumatoid arthritis genetics. In Cluster 1, molecular and transcriptomic studies consistently implicate cytokine dysregulation, particularly IL6, TNF-α, and JAK–STAT pathway components, as core mediators of inflammation and drug response5,31,33. These findings underpin the rationale for targeted therapies such as JAK inhibitors, yet cross-cohort transcriptomic analyses show inconsistent predictive signatures for methotrexate or TNF inhibitor efficacy. Cluster 2 highlights strong consensus on shared autoimmune susceptibility genes such as HLA-DRB1 and PADI4, which exhibit reproducible risk effects across ancestries19,23,46. However, variants such as PTPN22 display marked population-specific heterogeneity, strongly associated with rheumatoid arthritis in European and North American cohorts but weak or absent in East Asian and African populations21,45. These inconsistencies suggest allelic heterogeneity or gene–environment interactions shaping regional disease risk. In Cluster 3, HLA-DRB1 remains the most robust universal locus, whereas other loci such as STAT4 and CTLA4 show divergent significance across populations, as demonstrated by studies in Iraqi, Mexican, and Algerian cohorts46,47,53. Such discrepancies limit the transferability of polygenic risk scores and highlight the need for ancestry-inclusive GWAS. Finally, Cluster 4 highlights the translational challenges in linking functional polymorphisms to treatment response. Variants in VDR, TNF, and TP53 have been correlated with biologic response, remission, or disease activity in some studies, yet replication remains inconsistent and clinical utility uncertain49,50,52,55.
Collectively, these patterns illustrate a maturing yet heterogeneous field, where established immunogenetic mechanisms coexist with ancestry-specific associations and persistent barriers to clinical translation. This study has several limitations that warrant consideration. First, the analysis relied exclusively on Scopus as the data source. While Scopus provides broad coverage, reliable citation metadata, and strong compatibility with bibliometric tools13,14, the use of a single database may have excluded relevant publications indexed only in Web of Science or PubMed. Variations in indexing standards, author affiliations, and institutional naming conventions across databases also pose challenges for data harmonization. Second, our search strategy required the presence of inclusion search terms in article titles, which may have led to the omission of some relevant studies where these terms were reported only in abstracts or keywords. Third, the analysis was based on raw citation counts rather than normalized measures. As a result, some influential recent publications may be underrepresented in citation-based rankings. Fourth, the overlay visualization generated using VOSviewer is based on the average publication year. While this approach is useful for indicating the relative recency of research themes, it does not capture the frequency dynamics of keyword usage over time. Accordingly, our findings should be interpreted in light of these constraints.
CONCLUSIONS
Bibliometric analysis of rheumatoid arthritis and genetics research reveals a field marked by steady growth and evolving scientific priorities. From foundational breakthroughs in the late 20th century to the recent surge in interest in pharmacogenetics, epigenetics, and data-driven methodologies, the field has matured into a highly interdisciplinary domain. Thematic keyword clusters further reflect the field’s transition from traditional immunogenetics toward personalized medicine, complex comorbidity research, and computational genomics, signaling a future direction centered on precision healthcare and integrative approaches to disease understanding and management.
Research Gaps and Future Agenda
Although data-driven and integrative genomic approaches are increasingly shaping rheumatoid arthritis research, several knowledge gaps remain that limit the translation of genetic discoveries into clinical benefit. Current GWAS are still dominated by European ancestry cohorts, resulting in limited transferability of risk loci and polygenic risk scores to underrepresented populations5,8,20. Furthermore, functional follow-up studies integrating multi-omics layers such as epigenomic QTLs, single-cell transcriptomics, and proteomics remain sparse25,31,33. These gaps hinder the identification of causal variants and the mapping of gene regulatory networks underlying extra-articular manifestations such as interstitial lung disease or cardiovascular comorbidities3,39. Moreover, while polygenic risk scores and Mendelian randomization offer tools for prediction and causal inference, their clinical validation and incorporation into personalized treatment strategies remain nascent34,36,51.
Future research should prioritize the following questions to accelerate scientific and translational progress:
-
How can multi-ancestry GWAS improve fine-mapping accuracy and reveal population-specific risk variants in rheumatoid arthritis?
-
What role can multi-omics Mendelian randomization play in clarifying causal pathways linking genetic variants to extra-articular manifestations (e.g., lung fibrosis, atherosclerosis)?
-
How can integrative analyses of epigenomic QTLs and single-cell data refine our understanding of disease-relevant immune cell subtypes?
-
What strategies can enhance the clinical utility, equity, and calibration of polygenic risk scores across diverse ancestries?
-
How can predictive machine learning models be combined with genomic data to guide therapeutic response and drug repurposing?
Addressing these questions will be pivotal for transforming genetic discoveries into precision healthcare tools for rheumatoid arthritis.
ABBREVIATIONS
ACPA: anti-citrullinated protein antibody, anti-CCP: anti-cyclic citrullinated peptide, CKB: China Kadoorie Biobank, COVID-19: coronavirus disease 2019, CTLA4: cytotoxic T-lymphocyte-associated protein 4, DNA: deoxyribonucleic acid, GWAS: genome-wide association studies, HGP: Human Genome Project, HLA: Human Leukocyte Antigen, JAK-STAT: Janus kinase - signal transducer and activator of transcription, PTPN22: protein tyrosine phosphatase non-receptor type 22, QTLs: epigenomic quantitative trait loci, RFLP: restriction fragment length polymorphism, RNA: ribonucleic acid, SLE: systemic lupus erythematosus, SNP: single-nucleotide polymorphism, TNF: tumor necrosis factor, VDR: vitamin D receptor.
ACKNOWLEDGEMENTS
None.
AUTHORS’ CONTRIBUTIONS
(I) Conceptualization, methodology development, data curation, formal analysis, and drafting of the original manuscript: WNAZ (II) Administrative support, critical review, and manuscript editing: AW, WSWG, NDMS (III) Data acquisition, validation, and analysis: WNAZ, AW (IV) Final approval of the manuscript and accountability for the integrity of the work: All authors.
FUNDING
None.
AVAILABILITY OF DATA AND MATERIALS
Supplementary files, including the bibliometric dataset (.csv) and reference file (.ris), are available in Zenodo at
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Declaration of generative AI and AI-assisted technologies in the writing process
The authors declare that they have not used generative AI (a type of artificial intelligence technology that can produce various types of content including text, imagery, audio and synthetic data).
Competing interests
The authors declare that they have no competing interests.

