Non-Enzymatic Isolation of Stromal Vascular Fraction (SVF) And Adipose-Derived Stem Cells (ADSCs): A Systematic Methodological Review and Meta Analysis
- Reconstructive Sciences Unit, School of Medical Sciences, Health Campus, Universiti Sains Malaysia, 16150 Kota Bharu, Kelanta, Malaysia
- Department of Pathology, School of Medical Sciences, Health Campus, Universiti Sains Malaysia, Kubang Kerian, 16150, Kelantan, Malaysia
- Department of Medical Microbiology & Parasitology, School of Medical Sciences, Universiti Sains Malaysia Health Campus, Kubang Kerian 16150, Malaysia
- Reconstructive Sciences Unit, School of Medical Sciences, Health Campus, Universiti Sains Malaysia, 16150 Kota Bharu, Kelantan, Malaysia
Abstract
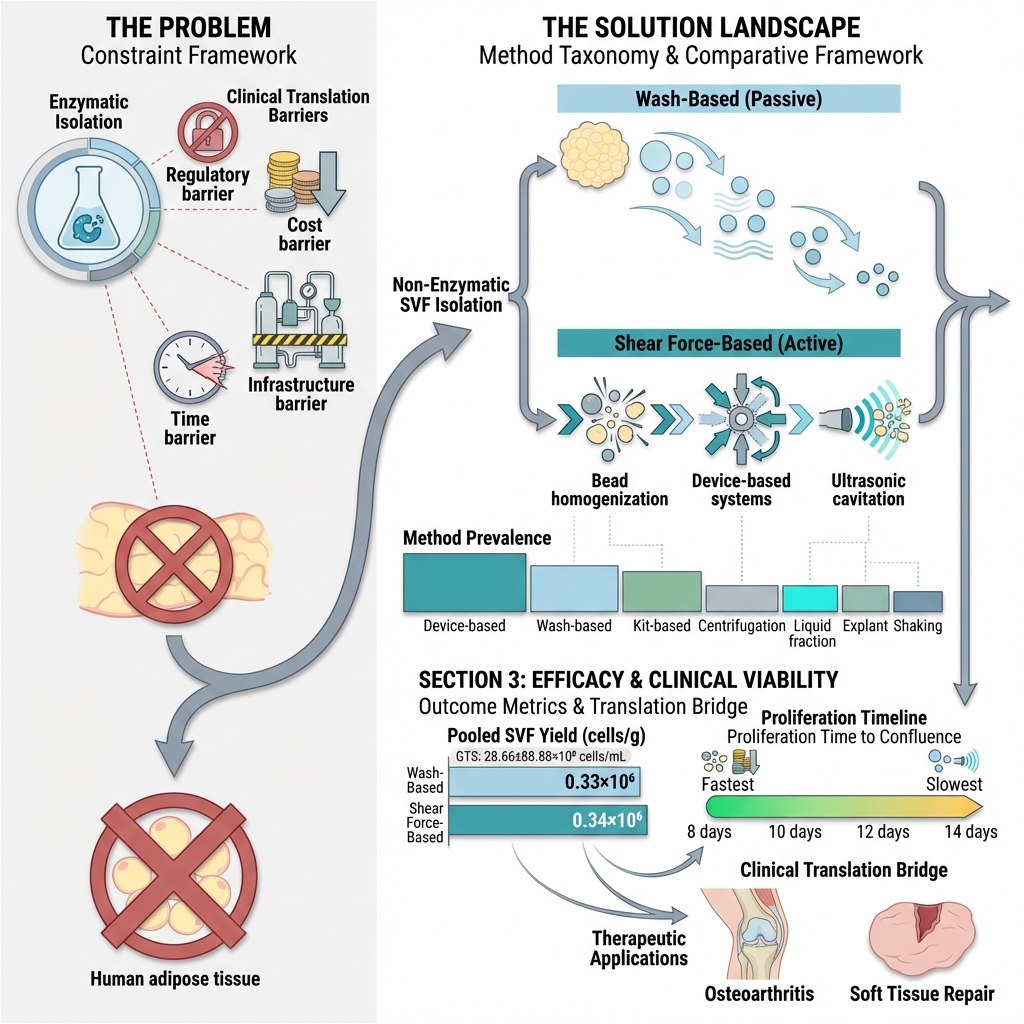
Non-enzymatic isolation of the stromal vascular fraction (SVF) has gained increasing clinical interest due to the regulatory, safety, and logistical constraints associated with enzymatic digestion. However, reported outcomes following their use remain heterogeneous, and direct quantitative comparisons are limited. This systematic review and exploratory meta-analysis aimed to compare SVF yield and in vitro proliferation outcomes across different non-enzymatic isolation techniques. A PRISMA-guided search of PubMed, Scopus, ScienceDirect, Google Scholar, and the Cochrane Library identified 17 eligible studies published up to May 2023. Device-based systems were the most frequently reported (35.3%), followed by wash-based methods (15%), kits (15%), centrifugation (12%), and other mechanical approaches. The pooled mean SVF yield was comparable between wash-based (0.33 × 10⁶ cells/g) and shear force–based techniques (0.34 × 10⁶ cells/g). The proliferative capacity varied across methods, with some mechanical and kit-based systems reaching confluence earlier than wash-based approaches. Substantial heterogeneity was observed across the included studies. These findings present clinically feasible alternatives for SVF recovery, demonstrating comparable mean cell yields across major mechanical categories. Increasing mechanical disruption does not necessarily enhance cell recovery. To enhance reproducibility and facilitate clinical translation, future research should prioritize standardized reporting frameworks and direct head-to-head scalability studies.
INTRODUCTION
Subcutaneous adipose tissue (AT) is widely used in plastic and reconstructive surgery, with applications including autologous fat grafting for breast augmentation, facial rejuvenation, scar revision, and soft tissue reconstruction 1,2. However, the extent of graft retention remains variable, with reported resorption rates ranging from 20% to 90% 1,2,3. Increasing evidence suggests that clinical outcomes depend not only on mature adipocytes but also on the stromal vascular fraction (SVF), which contains adipose-derived stem cells (ADSCs) and other regenerative cell populations 4,5,6,7,8. Consistent with this, SVF and ADSCs contribute to tissue repair through paracrine signaling, immunomodulation, and angiogenic support 9,10,11,12.
Effective therapeutic application requires disruption of the extracellular matrix (ECM) to release stromal cells. Historically, enzymatic digestion was introduced to facilitate ECM degradation and increase cell yield 13,14,15,16,17,18 (Figure 1).
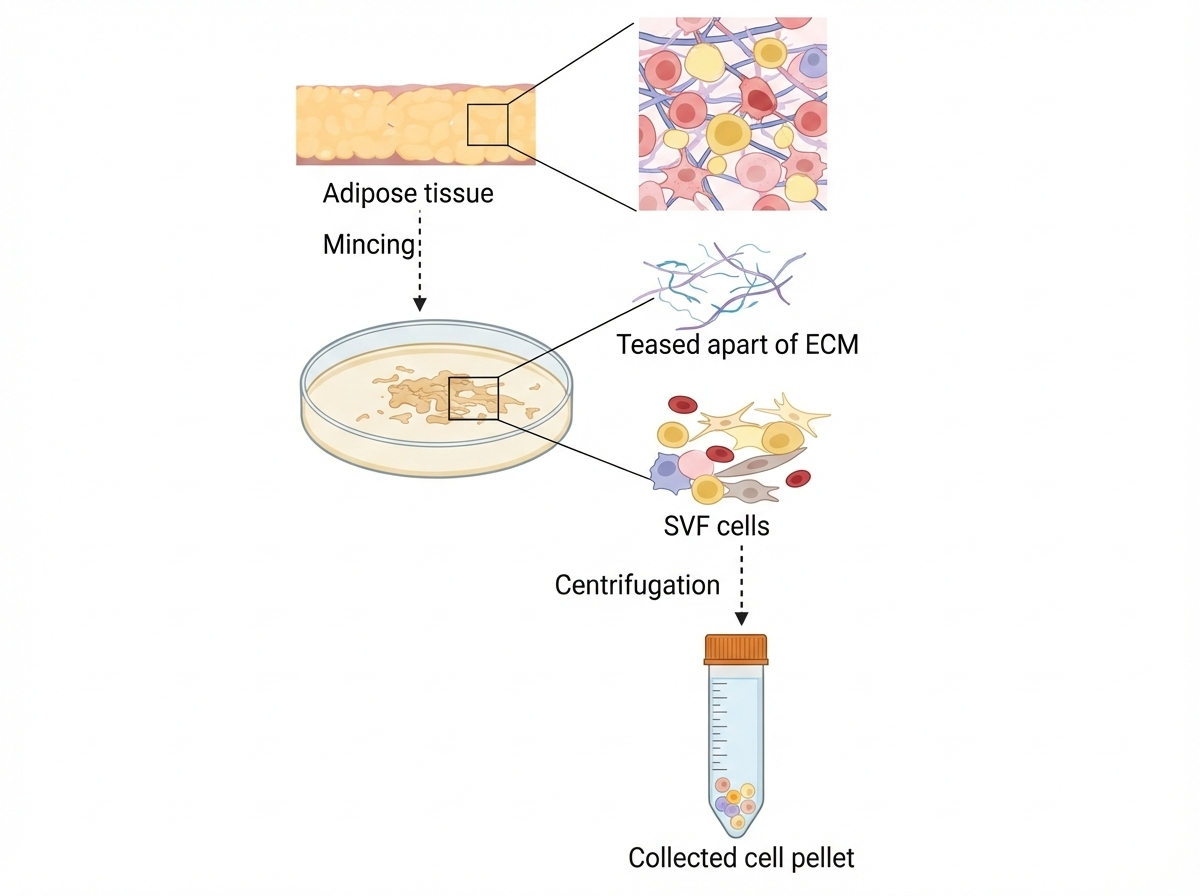
The principle of stromal vascular fraction cell isolation. Adipose tissue is minced to tear apart the peptide bonds which hold the cells together. Then the non-adipocyte cells called stromal vascular fraction (SVF) cells are concentrated at the bottom of the centrifuge tube. The figure is created in Biorender.com. No content was copied from copyrighted material.
Although enzymatic methods can achieve high cell recovery, they are associated with regulatory classification as “more than minimal manipulation,” increased cost, extended processing time, and the requirement for specialized laboratory infrastructure 19,20,21,22.
These regulatory and operational constraints have accelerated interest in non-enzymatic mechanical isolation strategies 23,24,25,26,27,28,29,30,31,32,33,34,35,36,37. Since their introduction in 2009, numerous enzyme-free techniques, including wash-based methods, shear force-based dissociation, and device-assisted systems, have been introduced to provide safer, faster, and more clinically adaptable alternatives 25.
Unlike prior reviews that describe mechanical approaches in a narrative manner, the present study systematically compares quantitative outcomes across non-enzymatic SVF isolation techniques. Specifically, this study evaluates SVF yield (cells/g), cell viability (%), and reported in vitro proliferation or confluence time. By categorizing techniques based on their underlying mechanical mechanisms and evaluating pooled mean yield, this review establishes quantitative benchmarks to guide methodological selection and clinical translation.
MATERIALS AND METHODS
This systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The review protocol was prospectively registered in PROSPERO (CRD42023493078).
Data sources and search strategies
A comprehensive literature search was performed to identify studies reporting non-enzymatic isolation of stromal vascular fraction (SVF) from human adipose tissue, published between January 2008 and May 2023. Searches were conducted in PubMed, Scopus, ScienceDirect, and the Cochrane Library. Google Scholar, a broad academic search engine rather than a curated bibliographic database, was used solely to extract all related studies. Embase and Web of Science were not searched due to substantial overlap in indexed biomedical literature with Scopus within the defined scope of this review.
Medical Subject Headings (MeSH) and free-text terms related to “stromal vascular fraction,” “adipose-derived stem cells,” “adipose tissue,” and “non-enzymatic isolation” were combined using Boolean operators (“AND,” “OR”). The PubMed search strategy was structured as follows: ((((((Human Adipose Tissue [Title/Abstract]) OR (Adipose-derived stromal cells [Title/Abstract])) OR (Stromal vascular fraction [Title/Abstract])) AND (Stem Cell Separation [Title/Abstract])) OR (Cell Separation [Title/Abstract])) OR (Cell Separation methods [Title/Abstract])) OR (Non-enzymatic Isolation systems [Title/Abstract])).
Equivalent search strings were adapted for other databases according to their indexing systems. The complete reproducible search strategy is provided in Supplementary Table S1 and Appendix 1.
Selection of studies
Study eligibility was determined using the PICOS framework (PRISMA-P 2016) (Appendix 2). Inclusion criteria were: (1) original studies published in English; (2) non-enzymatic isolation of SVF from human adipose tissue; (3) reporting of SVF or ADSC yield and/or viability; and (4) publication year ≥2008. Exclusion criteria included: (1) non-human adipose tissue studies; (2) enzymatic isolation methods; (3) absence of quantitative SVF outcomes; and (4) studies published before 2008.
Two reviewers independently screened titles, abstracts, and full texts. Disagreements were resolved through discussion and consensus by a third reviewer.
Data extraction
Extracted variables included donor characteristics, adipose tissue source, isolation technique, processing parameters, SVF/ADSC yield, cell viability, culture conditions, proliferation or confluence time, and limitations (Table 1).
Techniques of SVF cell isolation without enzyme
| Ref | Technique name/ device | Fat volume (g) | Process of SVF cell isolation | Result | Conclusion | |
|---|---|---|---|---|---|---|
| Confluence time | Cell yields (yield/g x 106/ml) | |||||
| Decantation | 27 | After the fat was allowed to sit for decantation in the syringes due to gravity, the middle layer and the three fundamental layers were noticed. | NA |
CD45-/CD34+ 2.8 ±2.7 % CD45-/CD31+ 3.8 ± 3.1% CD45-/CD105+ 2.9 ± 2.4 % | The optimal processing method for adipose tissue graft taking can end up being washed. Collagenase-digested SVF had higher numbers of ADSCs (60 %) than mechanically isolated SVF (6–13 %) and fewer hematopoietic origin cells (32 %) compared to mechanically isolated SVF (70–85 %). | |
| Washing | 45 | Washing three times in the same syringe in a 1:1 (vol/vol) ratio of normal saline produced a final volume of 35 ml of washed lipoaspirate or roughly two-thirds of the lipoaspirate that had been pre-washed. After that, the middle layer was gathered by observing the three fundamental levels. | NA |
CD45/CD34+ 4.3 ± 2.3 % CD45-/CD31+ 7.1 ± 3.6 % CD45-/CD105+ 4.7 ± 1.6 % | ||
| Centrifugation | 36 | Three fundamental layers were seen after being put in four sealed, 10-milliliter syringes and centrifuged for three minutes at a speed of 3000 revolutions per minute (rpm). The pellet was gathered at the bottom of the centrifuged sample as well as the middle layer. | NA |
CD45/CD34+ 2.3 ± 1.7 % CD45-/CD31+ 2.0 ± 0.7 % CD45-/CD105+ 2.1 ± 1.5 % | ||
| Wash | 4 | Following extensive washing with sterile PBS, the sample was placed in a culture plate with penicillin and streptomycin. Afterward, it was transferred to another plate with PBS, cut into small fragments, and then distributed in culture flasks for stem cell growth. On the third day, the culture medium was switched to DMEM supplemented with 10 % FBS. | 10 days | 1 × 107/ml (2.5) | ADSCs exhibited sustained long-term culture stability and proliferative potential. Additionally, the colony formation of ADSCs displayed a cell density-dependent increase over the same culture duration. | |
| Wash | 270 | Lipoaspirate was mixed with PBS and vigorously shaken to separate the tissue. The aqueous layer was collected in conical tubes, and the tissue was washed multiple times, saving each supernatant. The conical tubes were then centrifuged at 1200 rpm for 5 minutes at room temperature, resulting in the SVF pellet, which was resuspended in a stromal medium. | 7-21 days (average 13 days) | 2.5 × 104/ml (0.0000926) | The enzymatic method enhances cell yield and reduces culture time and passages when compared to the nonenzymatic approach. However, the nonenzymatic method remains crucial for processing large volumes of lipoaspirate in autologous applications. | |
| Rigenera® device | 3.6 | Within the Rigenera® capsule, lipoaspirate was blended with a full culture medium. The device was activated for durations of 30, 45, or 60 seconds. The resultant cell pellet was extracted from the capsule using a syringe, passed through a 70-µm nylon mesh, and subjected to centrifugation at 3000 rpm for 7 minutes. After discarding the supernatant, the cell pellet was enumerated. | 12 days | 2.320 ± 0.14 × 105 /ml (0.0644) | Enzymatic isolation provides higher cell yields than mechanical methods, yet Rigenera® must remain fast, safe, standardized, and autologous for clinical utility, with a 60-second treatment duration being particularly efficient. | |
|
Liquid Fraction Fast Sedimented | 7.2 | Centrifugation according to the Coleman technique (3 min at 3000 rpm e.g..1,811 g) and then left to decant for an additional 10 min. | 14 days | 0.31 ± 0.31 × 105 /mL (0.00431) | All cells obtained through tissue rupture were entirely gathered within the liquid portion of the lipoaspirates. After these mechanical methods, no disparity in cell proliferation capacity and vitality was observed in comparison to enzymatic dissociation employing a kit. | |
| Liquid Fraction Coleman | 7.2 | Centrifugation according to the Coleman technique (3 min at 3000 rpm e.g. 1,811 g). Cells were then collected from the fluid portion (2–3 mL) by centrifugation at 300 g for 5 minutes at room temperature. | 14 days | 1.76 ± 1.93 × 105 /mL (0.0244) | ||
| Wash fat Coleman | 7.2 | Following Coleman’s centrifugation technique (3 min at 3000 rpm or 1,811 g), cells were collected from the 2-3 mL liquid portion by further centrifugation at 300 g for 5 minutes at room temperature. The cell pellets were then suspended in red blood cell lysis buffer (160 mM NH4Cl) and incubated at 37°C for 5 minutes before filtering. | 14 days | 3.35 ± 4.76 × 105 /mL (0.0465) | ||
|
Liquid Fraction Long Sedimented | 7.2 | Centrifugation according to the Coleman technique (3 min at 3000 rpm e.g.,1,811 g) and then left to decant for an additional 20 min. | 14 days | 1.15 ± 1.68 × 105 /mL (0.016) | ||
|
Wash fat Sedimented | 2.7 | Adipose tissue was mixed with three volumes of PBS and incubated in a shaking incubator for 30 minutes with periodic shaking. Cells were separated from the adipocyte fraction via centrifugation at 300 g for 5 minutes at room temperature and subsequently suspended in red blood cell lysis buffer (160 mM NH4Cl) following 5 minutes of incubation at 37°C which later filtered through a 70 μm cell strainer (BD Biosciences). | 14 days | 3.98 ± 7.07 × 105 /mL (0.1474) | ||
| Liquid Fraction Kit | 2.7 | Recovered fat and fluid portions were separated by centrifugation of syringes at 300 g for 10 minutes. The cells from the liquid fraction were then counted. | 14 days | 3.04 ± 4.43 × 105/mL (0.1126) | ||
| Wash fat Kit | 2.7 | The fat and fluid portions were isolated by centrifuging the syringes at 300 g for 10 minutes which facilitated the division between broken fat (upper phase) and the liquid fraction (lower phase). To concentrate the cells, the liquid fraction (~1–2 mL) underwent further centrifugation at 300 g for 5 minutes. | 14 days | 2.43 ± 2.43 × 105/mL (0.09) | ||
| Explant | 450 | After washing the tissue with sterile PBS, it is finely chopped into small pieces (<1 mm) on the lid of a sterile 100 mm plate using a sterile needle and scalpel. The minced tissue is then transferred to a vacuum-gas plasma-treated culture plate, mixed with an equal volume of tissue culture media following the incubation until a sufficient number of cells adhere (more than 10 clusters of 15-25 cells each). Excess fluid is removed every 24 hours, and once enough cells are observed, the tissue is removed, and the culture process continues. | 7 days | NA | This method streamlines the isolation of SVF cells from adipose tissue, excluding harsh enzymes and centrifugation. Despite a lower initial yield, these cells exhibit robust in vitro proliferation, compensating for the initial lower yield. | |
| Gentle touch system (GTS) | 90 | Adipose tissue was mixed with PBS saline using injectors, with varied dilutions for indication-based protocols (IPs). Different IPs employed diluted or undiluted condensed tissue loaded into injectors on an atomizer disk and then cut into multiple micron sizes via back-and-forth movements. Following this, centrifugation at 1200g for 6 minutes yielded distinct layers. | 14 days | 28.66 ± 88.88 × 106 /ml (0.318) | Mechanical stromal cell transfer (MEST) presents a suitable technique for SVF isolation, with IPs 3 yielding the highest cell count, approximately 1.41 × 106/ml, from 8 ml of tissue while maintaining a cell viability of 93 %. | |
| ultrasonic cavitation | 54 | Adipose tissue is immersed in PBS, and the tissue composition is brought into contact with the ultrasonic probe of an ultrasonic cavitation device. The amplitude is initially set at approximately 50-100 % for 5-60 minutes and then gradually increased using a 14 mm ultrasonic rod after 5 minutes. Subsequently, the resulting solution is either allowed to settle over time or subjected to centrifugation. | NA | NA | Ultrasonication protocol results in about 10-fold more viable cells than collagenase protocol | |
| Lipogems | 90 | The device was preloaded with saline. The aspirated fat is then positioned within the device, which is kept vertically with five stainless steel marbles to facilitate the emulsification of oil residues. Once the solution inside the device appears clear, and the lipoaspirate turns yellow, halt the saline flow and invert the device (180°, gray cap facing up). The final Lipogems product is now prepared for the intended clinical or banking application. | 12 days | NA | This method demonstrates that the Lipogems product offers a straightforward approach for culturing ADSCs, whereas enzymatic processing in the same context necessitates significantly longer durations and additional handling. | |
| Lipogems device | 90 | The REAC apparatus, set at a 2.4 GHz "tissue optimization-regenerative protocol," was placed in a CO2 incubator with its electrodes immersed in the Lipogems culture medium, maintaining a 35 cm distance from the 2.4 GHz emitter. Electromagnetic measurements were conducted using a Tektronix model 2754p spectrum analyzer with an optimized receiving antenna. | 10 days | NA | REAC exposure did not harm ADSCs, and Lipogems-derived ADSCs showed increased transcription of vasculogenic genes in comparison to enzymatically dissociated cells | |
| Hy-Tissue SVF kit (Fidia Farmaceutici, Abano Terme, Italy) | 27 | With the bag containing fat positioned vertically, the lower section of the processing bag contained the Klein solution with some blood cells, which were then extracted. The fat tissue was massaged for 5 minutes and disaggregated with a small plastic rod, passing through a filter bag through manual massaging. The disaggregated tissue was collected with a syringe, centrifuged at 400 G for 10 minutes at room temperature, and subsequently resuspended in 1 mL of complete culture medium for cell counting. | 8 ± 2 days | 4.1 × 104 ± 2.0 × 104/mL (0.00152) | Despite a 3.2-fold reduction in SVF pellet number, compared to enzymatic digestion, the Hy-Tissue SVF system efficiently recovered up to 25 % of the SVF, indicating robust nucleated stromal cell separation. Moreover, there were no statistically significant differences in reaching confluence between SVF obtained via mechanical and enzymatic methods. | |
| Shaking | 72 | Tubes containing tissue were put into a vibrating shaker at 6000vib/min x 6 min. Then centrifuge at 1600rpm x 6min. All done under a laminar flow bench | NA | 1.25 x 105 /ml (0.001736) | The mechanical procedure described for isolating adipose-derived stem cells is a more straightforward, safer, cost-efficient, and expeditious alternative to traditional enzymatic methods currently employed. | |
| Ultrasonic cavitation | 45 | Initiate the laminar flow hood 3 minutes before the procedure. Connect the probe to the ultrasonic machine equipped with a 200W generator. Place the lipoaspirate into an ultrasonication tube for 10 minutes, then transfer the specimen from the machine into a sterile conical tube for filtration. Combine equal volumes of sodium chloride, perform a 3-minute centrifugation at 500 RCF, and finally, gather the stromal vascular fraction (SVF). | NA | 7.553x105/ml (0.01678) | This technique produces 10-fold more viable cells than comparable adipose tissue treated with collagenase. | |
| Homogenization with beads | 6.3 | Introduce 5 mL of 2 mm zirconium oxide beads into the fat sample, followed by placing the tube inside the Bullet Blender® Blue 50 instrument. Homogenize the sample on 1 speed for 3 minutes. Next, include approximately 10 mL of saline in the sample, and subsequently filter it through a 100 µm Steriflip filter for cell counting. | 8 ± 2 days | 2.3x106/ml (0.3651) | ||
| Indirect sonication | 54 | The tissue was positioned within a metal bowl submerged in water and subjected to sonication for approximately 30 minutes at an amplitude of about 100 and 200W. Following sonication, a 50/50 ratio of normal saline was introduced, and then the sample was used to collect pellets. | NA | 1.09x107/ml (0.20185) | ||
| Sonication cavitation | 0.9 | Blend for 10 seconds following immersion in a 50W sonication bath for 2 seconds. Subsequently, centrifuge at 900g for 10 minutes, resuspend the pellet in 150 mM ammonium chloride for 5 minutes, and centrifuge at 400g for 5 minutes. Finally, resuspend the pellets in DMEM + 20 % FBS and wash non-attached cells after 24 hours. | 14 days | 2.5 × 104/ml (0.02778) | The sonication-induced cavitation technique presents a potentially swift, secure, and economical means to isolate SVF and ADSCs at a substantial scale. The harvested cells have the potential for utilization in clinical trials or therapeutic applications. | |
| Centrifugation (CENT1 and CENT2) | NA | Spin the fat at speeds of 800g and then 1,2809g for 15 minutes each. Following the removal of the supernatant, incubate the pellet with 10 ml of red blood cell lysis buffer for 5 minutes at room temperature. Subsequently, centrifuge the mixture at 6009g for 10 minutes. After this, resuspend the pellets from all groups in the culture media. | NA | 2.5 × 104 | While viable cells were successfully obtained using the CENT1 and CENT2 protocols, they did not demonstrate proliferation even after 14 days of culture. | |
| Explant | 5 | Excess blood was eliminated by mixing with phosphate-buffered saline and settling for 5 minutes, separating the aqueous and fat phases. The fat was minced into 5 mm3 fragments in a petri dish, evenly distributed on a tissue culture-treated dish, with 1g of tissue per 100 mm dish. Explants were cultured in a complete medium. | 14 days | 5–8 × 105/ml (0.16) | Explant culture is often the preferred method for obtaining adipose-derived stem/stromal cells in tissue engineering and regenerative medicine, especially with limited starting material, due to its simplicity and cost-effectiveness. | |
| Centrifugation | NA | After decantation and centrifugation, the pellet accumulated at the bottom of the NEXT syringe was collected. | NA | NA | The pellet contained very few viable nucleated cells, and attempts to culture plastic adherent stem cells were unsuccessful. Consequently, the P1 protocol results were excluded from this study's analysis. | |
| Microlyzer | 10ml | The condensed adipose tissue (10 mL) underwent 31 passes through a 2400-micron microlyzer and 101 passes through a 600-micron microlyzer. The resulting microlyzed tissue was washed with saline and centrifuged at 400×g for 10 min. After washing, the pellet was manually aspirated. Following enzymatic digestion and microanalyzer disruption, the cell suspension was filtered through a 100 μm strainer to extract individual cells and small clusters while retaining their original matrix. | 2 × 107/ml (2.222) | Microlyzers, particularly in regeneration investigations, will allow quick and high-rate separation at the bedside. | ||
| Rigenera® (Human Brain Wave HBW, Turin, Italy) | NA | Two portions of the harvested fat of each patient were inserted into each device, according to each manufacturer’s instructions | NA | 1.4 x 106/ml | Mechanical techniques are effective in isolating SVF from adipose tissue with low differentiative capability but high immunomodulatory effect through cytokines and growth factors release. | |
| Lipogems® ortho kit (Lipogems International SpA, Milan, Italy) | 1.4 x 106/ml | |||||
| Vortexing and Centrifugation | 9 | Adipose tissue and 10 ml of phosphate-buffered saline were vibrated at 3200 rpm for 6 minutes and subsequently centrifuged at 558 g for 6 minutes. The mixture was filtered through a 100-µm sieve and centrifuged at 558 g for 10 minutes. Then the supernatant was discarded, and the pellet was resuspended in a complete culture medium. | NA | 54.53 ± 7.55 % | Enzymatic isolation yielded a higher number of viable SVF cells and colony-forming units compared to the non-enzymatic method, but the difference in colony-forming units was not statistically significant. | |
| Dissociation by Intersyringe Processing. | 9 | Ten grams of washed adipose tissue were mechanically emulsified through 30 passes between Luer-Lok–connected 10-cc syringes, resulting in a whitish, almost liquid adipose tissue. After filtration through a 100-µm filter, the cell suspension was centrifuged at 558 g for 10 minutes, and the pellet was resuspended in a complete medium. The stromal vascular fraction, representing the adipose-derived stromal cell concentration, was determined using a hemocytometer. | 45.53 ± 3.49 % | Enzymatic digestion results in increased stromal cell yields, whereas the mechanical inter-syringe method provides a swift and efficient means to isolate stromal cells with stemness and immunosuppressive characteristics, streamlining the acquisition of enriched stromal vascular fractions for healthcare providers. | ||
When outcomes were reported in heterogeneous units (e.g., total cell count or cells/mL), values were normalized to cells per gram of adipose tissue by dividing the total reported cell count by the corresponding tissue mass.
For analytical purposes, isolation techniques were categorized into two groups according to their underlying processing mechanism: wash-based methods and shear force–based mechanical dissociation methods. Wash-based techniques rely primarily on dilution, rinsing, and centrifugation to separate SVF without active tissue disruption. In contrast, shear force–based approaches apply mechanical stress to fragment the extracellular matrix and enhance stromal cell release. The latter group included Rigenera®, Gentle Touch System (GTS), ultrasonic cavitation, homogenization with beads, indirect sonication, and centrifugation-based mechanical protocols (CENT1 and CENT2).
This classification facilitated a mechanism-oriented comparison of SVF yield between passive separation strategies (wash-based methods) and active mechanical dissociation techniques (shear force–based approaches).
Data synthesis
Outcomes were synthesized under two primary domains: (1) SVF cell yield and (2) in vitro proliferation or confluence time.
Because several studies did not consistently report seeding density, culture medium composition, or culture surface area, factors that directly influence proliferation, quantitative pooling of confluence outcomes was not performed. These results were therefore interpreted descriptively to reduce confounding and preserve methodological validity.
Risk-of-bias assessment
Risk of bias was assessed using the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for quasi-experimental and in vitro studies. Two reviewers independently evaluated each study across domains including selection bias, exposure measurement, outcome assessment, reporting bias, and statistical analysis. Each item was rated as low or high risk of bias, and results are summarized in Figure 2.
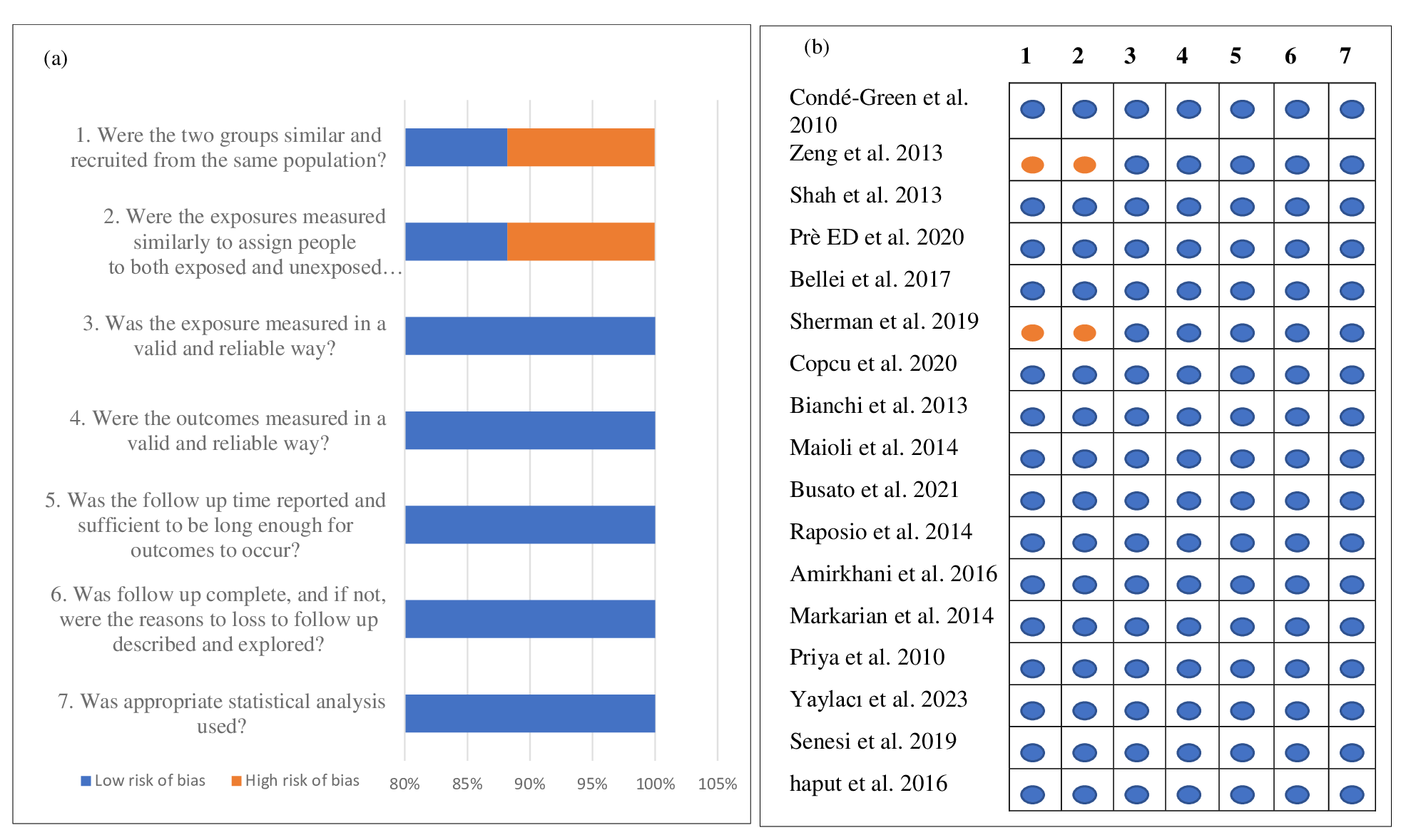
Risk of bias for studies -(a) graph and (b) summary; = yes/ low risk of bias and = no/ high risk of bias.
Detailed item-level assessments and scoring for each included study are provided in Appendix 3 (Supplementary File) to enhance transparency. Although the included studies were predominantly small in vitro experiments, most appraisal domains were rated as low risk because essential methodological criteria were adequately described.
STATISTICAL ANALYSIS
For outcomes reported in three or more studies, exploratory random-effects meta-analysis was performed to estimate pooled mean values with 95% confidence intervals. Statistical heterogeneity was assessed using the I² statistic. Statistical analyses were conducted using RStudio (Posit version 2026.01.0+392).
RESULTS
The PRISMA flow diagram summarizes the literature search and study selection process. Database searches yielded 440 records from PubMed, 80 from Cochrane, 14 from Scopus, 451 from ScienceDirect, and 479 from Google Scholar. After removal of duplicates, 984 unique records were screened. Any discrepancies in preliminary record counts were addressed during full-text review and are clarified in the updated PRISMA diagram. Following full-text assessment, 17 studies met the predefined eligibility criteria and were included in the final analysis (Figure 2).
Key characteristics of the included studies are summarized in Appendix 4. The studies were conducted in multiple countries, with a predominance of studies from Italy, and were published in journals indexed in Web of Science, PubMed, and Scopus. Cell Transplantation, Scientific Reports, and Plastic and Reconstructive Surgery each contributed two studies. Across all studies, female donors predominated (n = 99), with reported ages ranging from 10 to 74 years.
Efficacy of SVF Isolation Techniques Without Enzymes
All 17 studies employed non-enzymatic approaches for in vitro SVF analysis (Table 1) . Mechanical isolation strategies included decantation, washing, centrifugation, shaking, intersyringe processing, liquid fraction sedimentation, homogenization with beads, explant culture, and device-based systems. The Gentle Touch System (GTS) demonstrated the highest total SVF yield (28.66 ± 88.88 × 10⁶ cells/mL), followed by Microlyzer (2 × 10⁷ cells/mL), indirect/ultrasonic cavitation (1.09 × 10⁷ – 7.553 × 10⁵ cells/mL), homogenization with beads (2.3 × 10⁶ cells/mL), Lipogems® (1.4 × 10⁶ cells/mL), Rigenera® (2.320 ± 0.14 × 10⁵ cells/mL), and explant culture (5–8 × 10⁵ cells/mL) . However, substantial variation in reported yields likely reflects differences in processing parameters, tissue volume, reporting units, and study frequency rather than true biological superiority of specific techniques. The distribution of isolation approaches was as follows: device-based systems (35.3%)26, wash-based methods (15%)26,27,28,29, kits (15%), centrifugation (12%)34,39, liquid fraction techniques (12%)29, explant culture (12%), shaking (12%)40,41, decantation (5%), bead homogenization (5%), and intersyringe processing (5%)⁴¹ (Figure 3, details in Appendix 5) .
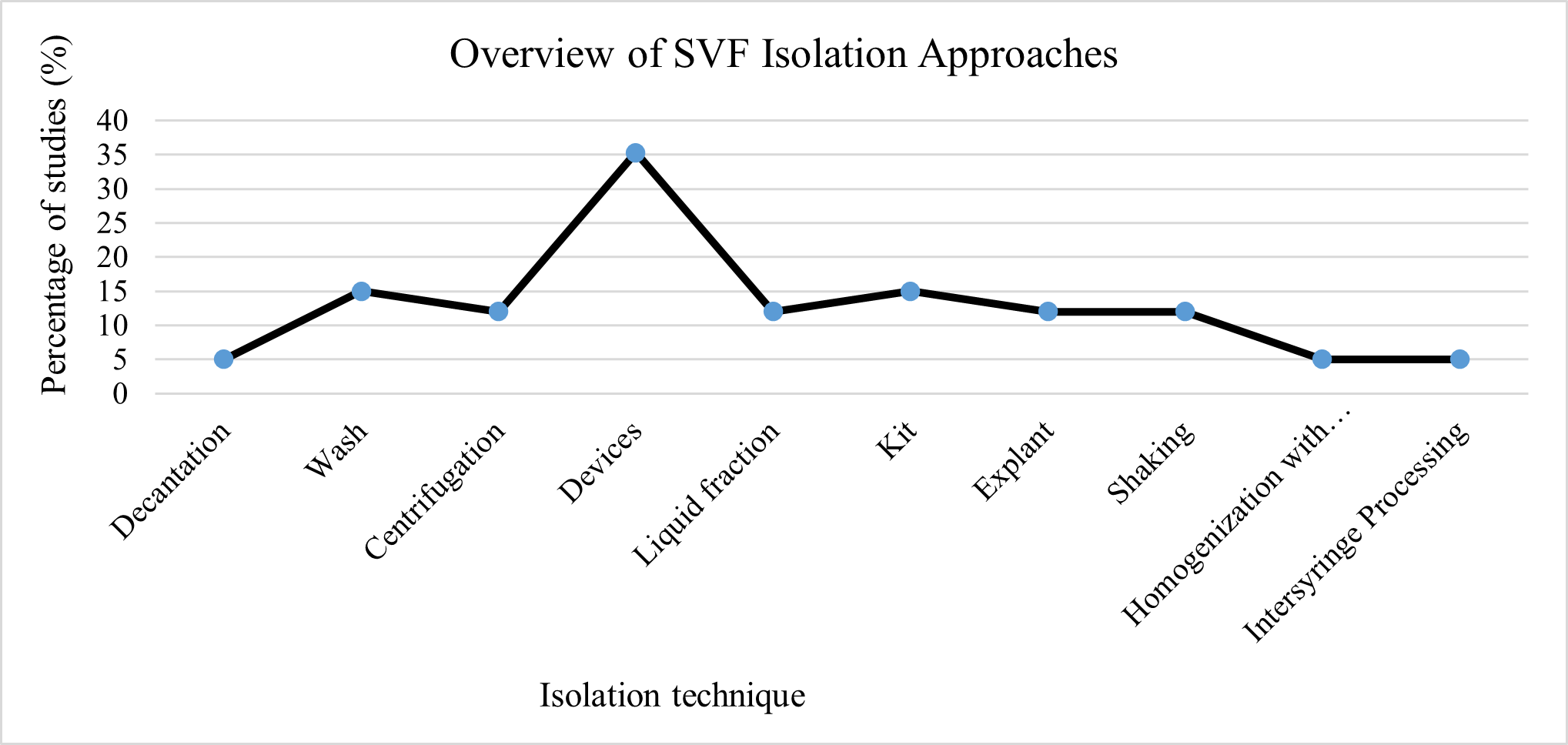
Use of different methods for isolating SVF containing adipose-derived stem cells (ADSCs).
To enable a standardized comparison across studies, SVF yields were normalized to cells per gram of adipose tissue and analyzed using an exploratory pooled mean approach. Techniques were categorized as wash-based ( passive separation ) or shear force–based mechanical dissociation ( active disruption ) methods.
Pooled Mean Analysis of SVF Yield Per Gram of Adipose Tissue
The pooled mean SVF yield was 0.33 × 10⁶ cells/g for wash-based techniques and 0.34 × 10⁶ cells/g for shear force–based techniques (Figures 4 and 5) . Despite considerable inter-study heterogeneity, the pooled estimates were comparable between categories (Appendix 6) . Given the variability in donor characteristics, processed fat volumes, mechanical intensity, and downstream handling protocols, pooled results were interpreted descriptively, not inferentially.
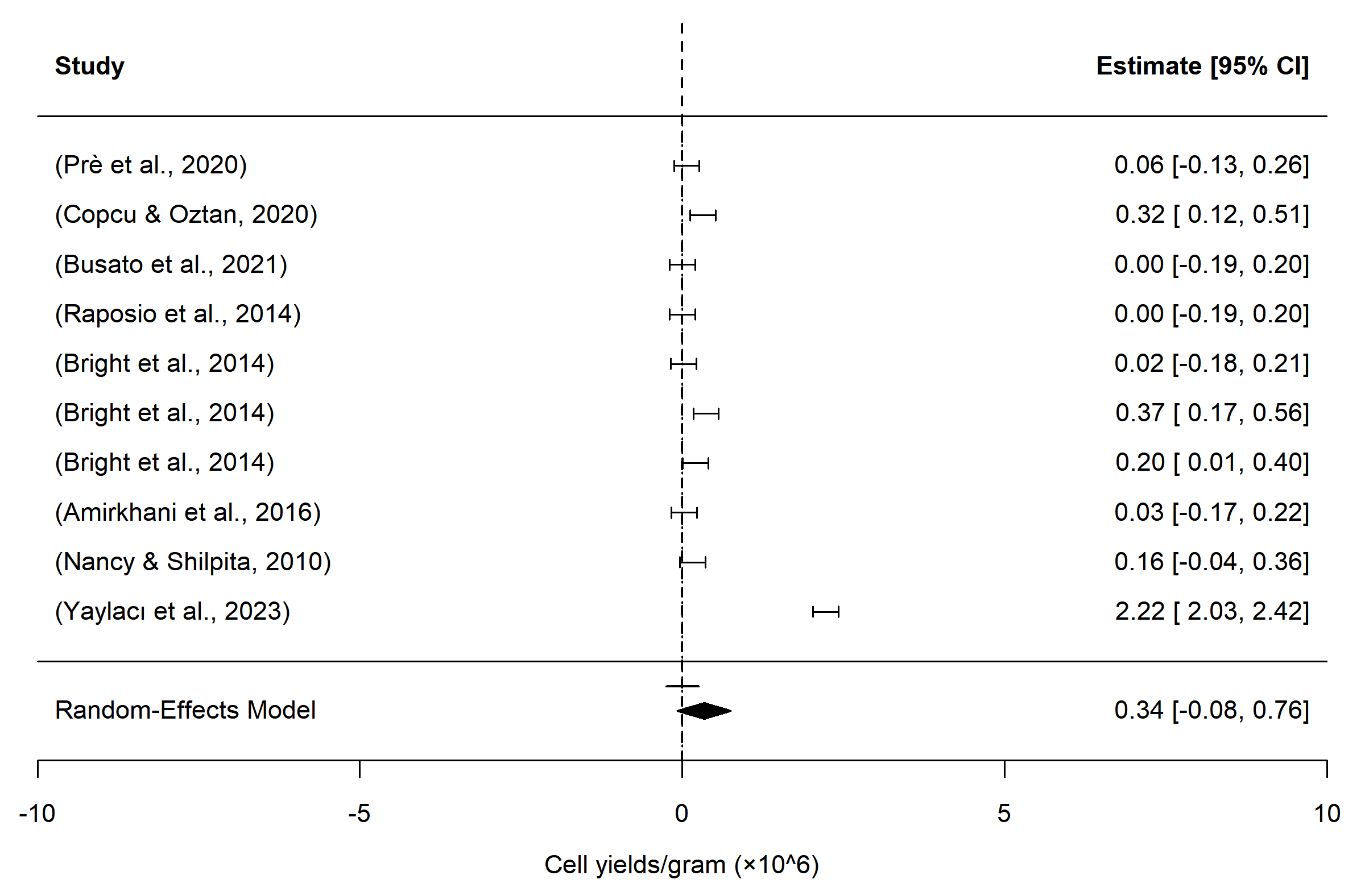
Forest plot showing pooled mean stromal vascular fraction (SVF) yield (cells/g adipose tissue) from shear force–based non-enzymatic isolation methods, with squares representing individual study estimates (95% CI) and the diamond indicating the random-effects pooled mean.
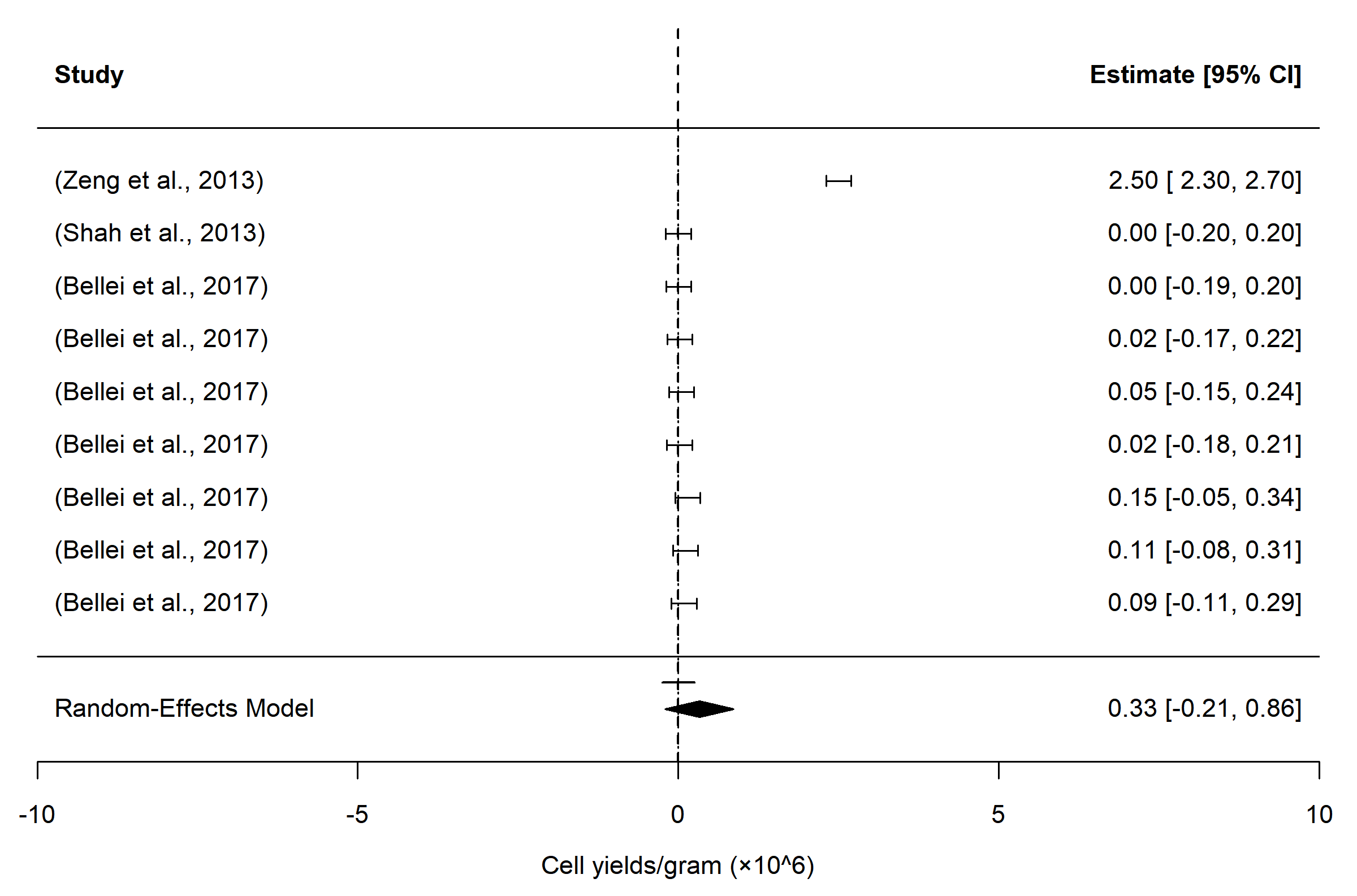
Forest plot illustrating pooled mean stromal vascular fraction (SVF) yield (cells/g adipose tissue) from wash-based non-enzymatic isolation methods, with squares representing individual study estimates (95% CI) and the diamond indicating the random-effects pooled mean.
Proliferation behavior also differed across techniques. Time to confluence varied according to both isolation method and initial seeding density, ranging from 8 days for homogenization with beads (2.3 × 10⁶ cells) and the Hy-Tissue SVF kit (4.1 × 10⁴ cells), to 9 days for liquid fraction methods (1.56 × 10⁵ cells), 11 days for Lipogems® (1.4 × 10⁶ cells) and explant culture (6.5 × 10⁵ cells), and up to 13–14 days for wash-based methods and ultrasonic cavitation, including the Gentle Touch System (Table 1) .
DISCUSSION
The clinical outcomes of stromal vascular fraction (SVF) and adipose-derived stem cells (ADSCs) are strongly influenced by the chosen isolation method, as different techniques can significantly affect cell yield, viability, phenotypic composition, functional capacity, and overall therapeutic efficacy 42,43,44. Although enzymatic digestion, commonly using collagenase, offers efficient tissue dissociation and high cell yield, its clinical translation is constrained by regulatory concerns regarding substantial manipulation, risks of residual enzyme activity, stringent sterility requirements, higher costs, longer processing time, and the need for specialized laboratory infrastructure 19,29,36,45. These limitations have restricted routine point-of-care use and have accelerated interest in non-enzymatic mechanical isolation as a more practical and regulatory-compliant alternative. In this context, the present review focuses exclusively on non-enzymatic SVF isolation techniques, providing a comprehensive methodological synthesis of mechanical strategies, comparative performance outcomes, and translational feasibility—an area that remains fragmented and insufficiently standardized in the existing literature.
Overall, non-enzymatic SVF isolation methods yield sufficient cell numbers for common clinical applications, such as treating osteoarthritis, chronic wounds, and facilitating soft tissue repair, where typical therapeutic doses are 1–10 × 10⁶ cells per injection. Higher-yielding device-based systems reliably meet these requirements, while moderate-yield methods may be suitable for smaller-volume or adjunctive treatments. Even lower-yield techniques can serve supportive roles, as SVF contains a heterogeneous mix of regenerative cells including mesenchymal stem cells (ADSCs), endothelial progenitors, and immune -modulatory populations that can enhance tissue repair and modulate inflammation even at lower cell counts.
In terms of technique, device-based systems were the most frequently reported, followed by wash-based methods, kits, centrifugation, liquid fraction techniques, explant culture, shaking, decantation, bead homogenization, and intersyringe processing. The predominance of device-based systems reflects a shift toward more standardized and reproducible mechanical platforms, likely driven by clinical demands for closed processing and regulatory compliance. Nevertheless, the limited representation and lack of direct comparisons for simpler mechanical techniques make it difficult to draw definitive conclusions regarding their relative efficacy.
The pooled mean analysis of SVF yield per gram of adipose tissue provides a useful quantitative benchmark for non-enzymatic isolation techniques, highlighting that wash-based and shear force–based methods achieve broadly comparable average recovery. Specifically, the similarity in pooled yields (0.33 × 10⁶ cells/g vs. 0.34 × 10⁶ cells/g) suggests that, at a gross level, both categories can generate sufficient SVF for many clinical applications, despite differences in mechanical processing approaches. This finding reinforces the notion that high SVF yields are not exclusive to one specific method and that multiple non-enzymatic strategies can potentially meet therapeutic thresholds for regenerative cell therapies.
However, the pooled mean also masks substantial inter-study heterogeneity arising from multiple sources, including donor factors (age, BMI, metabolic status), adipose tissue harvest site, processed fat volumes, mechanical intensity, device specifications, and post-isolation handling procedures. Such variability emphasizes that SVF yield alone may be insufficient to define clinical efficacy, highlighting the importance of optimizing isolation parameters and standardizing mechanical techniques to maximize not only cell recovery but also consistency and reproducibility across donors and clinical settings.
Despite this variability, the pooled estimates provide new insights by offering a reference point for expected SVF recovery in enzyme-free protocols, which can guide clinical planning, protocol optimization, and regulatory assessment. For example, knowing the average yield per gram allows clinicians and researchers to estimate the volume of adipose tissue required to achieve a target therapeutic cell dose for conditions such as osteoarthritis, chronic wounds, or soft tissue repair. From a research perspective, this benchmark enables future cross-study comparisons and meta-analytic integration, helping to identify which non-enzymatic strategies consistently yield substantial numbers of SVF cells.
Beyond yield, the functional behavior of the isolated cells is equally important. The proliferation behavior of SVF cells varied notably across non-enzymatic isolation techniques, reflecting differences in both initial cell yield and method-specific impacts on cell viability and functionality. Techniques such as bead homogenization and the Hy-Tissue SVF kit reached confluence faster, likely due to higher initial cell numbers and reduced mechanical stress, whereas wash-based methods, ultrasonic cavitation, and the Gentle Touch System required longer times. Other approaches, including liquid fraction methods, Lipogems®, and explant culture, showed moderate proliferation rates. These findings indicate that the isolation method can influence not only cell yield but also proliferative capacity, which is critical for in vitro expansion, quality control, and preparation for clinical applications. Understanding proliferative kinetics also depends on initial cell seeding density, which was inconsistently reported across studies. Even without complete seeding data, the observed trends provide valuable guidance for future research by highlighting the relative growth potential of SVF obtained via different non-enzymatic methods and emphasizing the need for standardized reporting of both cell numbers and proliferation metrics to optimize experimental protocols and support translation to future clinical applications.
LIMITATIONS OF THIS STUDY
This review has several limitations. Substantial heterogeneity existed across included studies with respect to donor characteristics, adipose tissue source, processed fat volume, mechanical intensity, and culture conditions, limiting direct comparability. Publication bias may be present, as studies with favorable yield or viability outcomes are more likely to be published. Additionally, inclusion was restricted to English-language publications, introducing potential language bias and limiting representation of relevant data from non-English sources. The exploratory pooled mean analysis was constrained by small sample sizes, inconsistent reporting of variance measures, and high statistical heterogeneity. Furthermore, limited head-to-head comparisons between techniques reduce confidence in any claims of comparative superiority.
STRENGTHS OF THIS STUDY
This study’s strengths include a systematic search strategy and a comprehensive synthesis of non-enzymatic SVF isolation techniques with quantitative yield and proliferation data. By highlighting both isolation efficiency and post-isolation culture behavior, it provides clinically relevant guidance for method selection and protocol refinement. Future research should prioritize standardized reporting of SVF and ADSC viability, donor characteristics, seeding density, and culture conditions; head-to-head comparisons of functional quality, reproducibility, scalability, and GMP-aligned clinical feasibility; and evaluation of functional outcomes such as immunophenotype, differentiation capacity, and secretome profile to enhance translational relevance.
CONCLUSION
Non-enzymatic stromal vascular fraction (SVF) isolation techniques provide clinically feasible alternatives to enzymatic digestion, particularly in settings where regulatory compliance, cost, and procedural simplicity are critical. No single method demonstrates universal superiority; rather, each involves trade-offs between yield, viability, proliferation, and operational complexity. The comparable pooled mean yield observed between wash-based and shear force–based approaches suggests that increasing mechanical disruption does not necessarily enhance recovery. Future research should prioritize standardized reporting and direct comparative scalability studies to optimize reproducibility and facilitate regulatory-aligned clinical translation.
ABBREVIATIONS
ADSCs: Adipose-Derived Stem Cells; AT: Adipose Tissue; BMI: Body Mass Index; CENT1: Centrifugation Protocol 1; CENT2: Centrifugation Protocol 2; DMEM: Dulbecco's Modified Eagle Medium; ECM: Extracellular Matrix; FBS: Fetal Bovine Serum; g: Relative centrifugal force (times gravity); GMP: Good Manufacturing Practice; GTS: Gentle Touch System; IPs: Indication-based protocols; JBI: Joanna Briggs Institute; MeSH: Medical Subject Headings; MEST: Mechanical Stromal Cell Transfer; PBS: Phosphate-Buffered Saline; PICOS: Population, Intervention, Comparison, Outcome, Study design; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; PROSPERO: International Prospective Register of Systematic Reviews; REAC: Radio Electric Asymmetric Conveyer; rpm: revolutions per minute; SVF: Stromal Vascular Fraction.
ACKNOWLEDGMENTS
We thank all the staff of the PRS unit.
AUTHOR’S CONTRIBUTIONS
The first draft of the manuscript was written by N.A.M.N. and S.F. then the manuscript was reviewed by S.H.S. and W.A.W.S. All authors declared no potential conflicts of interest concerning the research, authorship, and publication of this article.
FUNDING
This work was funded by the Fundamental Research Grant Scheme (Grant Number: FRGS/1/2020/SKK0/USM/03/12) from the Ministry of Higher Education, Malaysia.
AVAILABILITY OF DATA AND MATERIALS
This study is a methodological systematic review. It involved the compilation and analysis of data extracted from previously published studies.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
CONSENT FOR PUBLICATION
Not applicable.
DECLARATION OF GENERATIVE AI AND AI-ASSISTED TECHNOLOGIES IN THE WRITING PROCESS
During the preparation of this work, the authors wrote, edited, and reviewed the content entirely themselves and take full responsibility for the content of the publication.
COMPETING INTERESTS
The authors declare that they have no competing interests.

