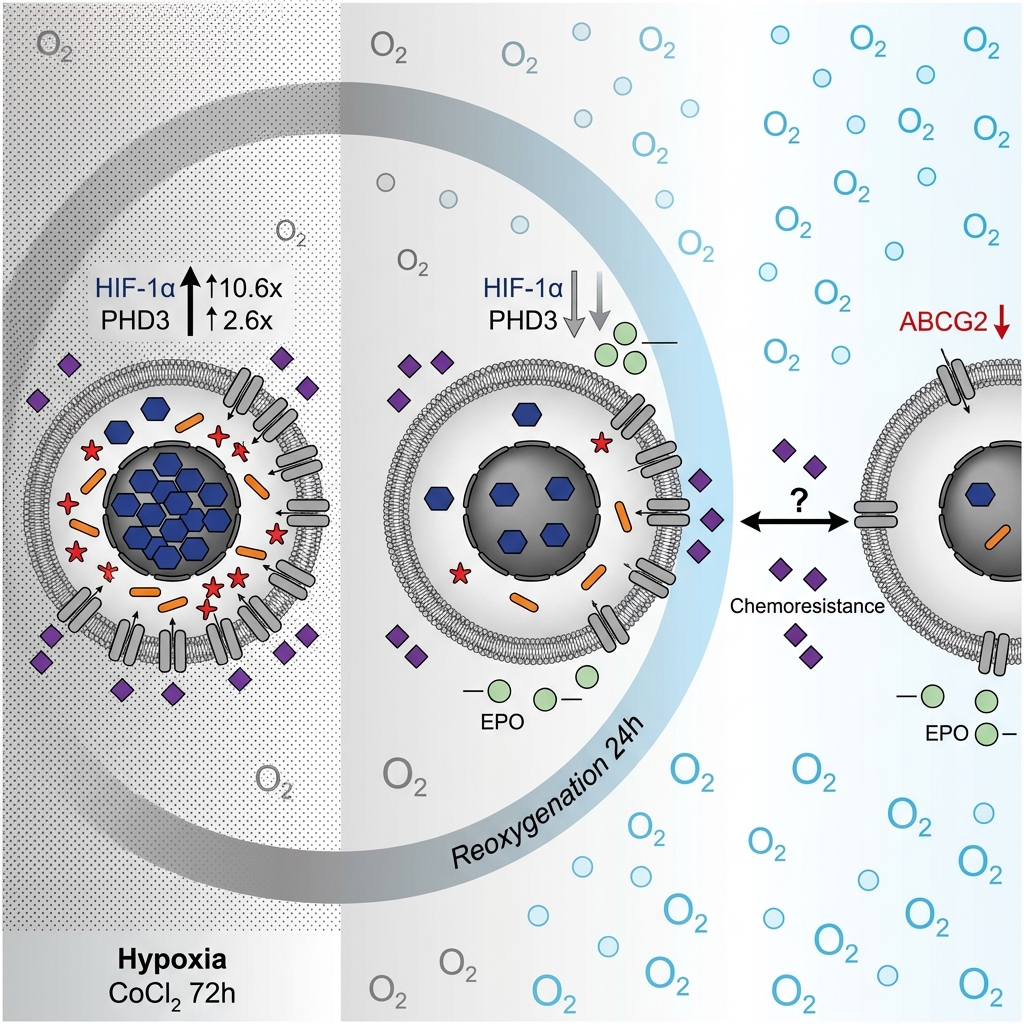
Impact of reoxygenation on Prolyl hydroxylase domain 3 expression and cisplatin sensitivity in breast cancer stem cells after prolonged hypoxia
- Stem Cell Institute, University of Science, Ho Chi Minh City, Viet Nam
- Viet Nam National University Ho Chi Minh City, Viet Nam
Abstract
Background: Targeting cancer stem cells (CSCs) within the tumor microenvironment has emerged as a critical focus in the development of novel oncological therapies. CSCs are frequently subjected to prolonged hypoxic conditions; while subsequent reoxygenation has been shown to significantly enhance the efficacy of radiotherapy across various cancer cell lines, its impact on chemotherapeutic resistance remains poorly understood.
Methods: In this study, an in vitro model was utilized where breast cancer stem cells were treated with cobalt chloride (CoCl₂) for 72 hours to induce hypoxia, followed by a 24-hour incubation in CoCl₂-free medium to simulate rapid reoxygenation. Alterations in hypoxia-related gene expression were quantified using RT-qPCR, and changes in BCSC chemoresistance were evaluated via alamarBlue-based IC50 assays.
Results: Both prolonged hypoxia and rapid reoxygenation significantly modulated the expression of the hypoxia-responsive gene HIF-1α and its regulatory target, PHD3. Notably, the expression of the drug efflux transporter ABCG2 was markedly downregulated upon reoxygenation following hypoxic preconditioning. Despite this molecular alteration, the associated reduction in cisplatin resistance did not reach statistical significance.
Conclusion: Collectively, these findings provide preliminary evidence that integrating oxygen-dependent therapeutics or enhancing oxygen delivery to hypoxic tumor cells influences resistance-associated gene expression, though further optimization is needed to significantly improve chemotherapy efficacy.
Introduction
In breast cancer, an increasing body of evidence highlights the critical role of cancer stem cells (CSCs) in driving tumor drug resistance1. Breast CSCs (BCSCs) are typically identified and isolated based on their distinct CD44⁺/CD24⁻/low surface marker phenotype2. While various intrinsic mechanisms underlying BCSC therapy resistance have been characterized, the specific contributions of the tumor microenvironment (TME) to treatment failure remain incompletely understood.
Hypoxia is a hallmark feature of the TME3, and accumulating literature demonstrates a strong association between hypoxic conditions and enhanced therapeutic resistance in CSCs4. Consequently, the development of robust in vitro hypoxic culture models for CSCs is imperative for the accurate assessment of their chemoresistance profiles. A prevalent method for simulating hypoxia involves treating cells with cobalt chloride (CoCl₂)5,6. Mechanistically, CoCl₂ acts to stabilize hypoxia-inducible factors (HIFs) by inhibiting the activity of prolyl hydroxylase domain (PHD) enzymes across various cancer cell lines7. This inhibition leads to the intracellular accumulation of HIFs, thereby triggering cellular responses that effectively recapitulate a hypoxic state8.
The HIF family consists of three recognized members (HIF-1, HIF-2, and HIF-3). Each functions as a heterodimer comprising an oxygen-labile α subunit (HIF-1α, HIF-2α, or HIF-3α)—which undergoes rapid degradation under normoxic conditions—and a constitutively expressed β subunit, also known as ARNT9. Extensive research demonstrates that HIF expression, particularly that of HIF-1, orchestrates numerous physiological and pathological pathways in both normal and malignant tissues10,11. Following hypoxia-induced activation, HIF acts as a transcription factor to directly promote angiogenesis and hematopoiesis by upregulating vascular endothelial growth factor (VEGF)12,13 and erythropoietin (EPO)14,15, respectively. Furthermore, HIF has been implicated in augmenting the drug resistance of breast cancer cells under hypoxic stress through the upregulation of the ATP-binding cassette subfamily G member 2 (ABCG2)16,17. Therefore, elucidating the comprehensive regulatory mechanisms governing HIF is crucial for effectively targeting these diverse, HIF-dependent cellular processes.
Prolyl hydroxylase domain (PHD) enzymes serve as primary regulators of HIF stability. Belonging to the prolyl-4-hydroxylase (P4H) protein family, PHD proteins execute post-translational modifications via the hydroxylation of prolyl residues on target proteins, thereby influencing a wide array of physiological processes in metazoans18,19,20. In humans, three principal isoforms exist: PHD1, PHD2, and PHD321. While substantial evidence characterizes PHD3 as a tumor suppressor whose inactivation bolsters cancer cell proliferation under hypoxia22, conflicting reports suggest that hypoxia-induced PHD3 expression may actively contribute to tumor initiation and progression23. Interestingly, PHD3 retains its functionality under both normoxic and prolonged hypoxic conditions24. This sustained activity is driven by a HIF-α–mediated positive feedback loop, wherein HIF-α binds to hypoxia response element (HRE) sequences within the PHD3 regulatory region25. Collectively, these dynamic characteristics position PHD3 as a highly relevant target in hypoxia-centric cancer research.
Beyond hypoxia, the subsequent restoration of oxygen levels—reoxygenation—has been shown to profoundly impact the trajectory of hypoxic cancer cells. For instance, the sequence of hypoxia followed by reoxygenation induces epithelial-mesenchymal transition (EMT) in colon cancer cells via an NF-κB-dependent signaling pathway26. Similarly, in colorectal cancer models, exposure to either extended hypoxia or subsequent reoxygenation stimulates cellular proliferation and confers durable resistance to apoptosis27. Although it is evident that both hypoxic and reoxygenation phases dictate cancer cell survival, the precise molecular mechanisms and downstream signaling networks governing these adaptations remain incompletely defined.
To bridge this knowledge gap, the present study utilizes a CoCl₂-induced hypoxia model in BCSCs to evaluate the expression dynamics of PHD3 and associated hypoxia-responsive genes. Ultimately, this research aims to determine how these molecular alterations influence chemotherapeutic resistance following periods of prolonged hypoxia and subsequent rapid reoxygenation.
Materials and Methods
Cell Culture
The Vietnamese-derived breast cancer cell line VNBRCA1, originally established in a previous study28, was obtained from the Cell Bank of the Stem Cell Institute at the University of Science, Vietnam National University Ho Chi Minh City (VNU-HCM). Cells were cultured in Dulbecco's Modified Eagle Medium/Nutrient Mixture F-12 (DMEM/F12; Gibco, USA), supplemented with 10% fetal bovine serum (FBS; Sigma-Aldrich, Germany) and a 1% antibiotic solution (Gibco, USA). The cultures were maintained under normoxic conditions at 37°C in a humidified incubator containing 5% CO₂. The culture medium was replenished every 48 hours. Upon reaching 70–80% confluence in culture flasks (SPL, Korea), the cells were dissociated using TrypLE Express (1X; Gibco, USA). The resulting cell suspension was centrifuged at 500 × g for 5 minutes, and the cell pellet was reseeded into new culture flasks for continued expansion.
Isolation of Breast Cancer Stem Cells (BCSCs) via Flow Cytometry
VNBRCA1 cells were expanded until the total yield reached approximately 2–3 × 10⁷ cells, at which point they were harvested for flow cytometric sorting. The cells were centrifuged at 500 × g for 5 minutes, the supernatant was discarded, and the pellet was resuspended in 210 μL of phosphate-buffered saline (PBS). For the unstained control, a 10 μL aliquot of this suspension was transferred to a 1.5-mL microcentrifuge tube containing 300 μL of PBS. The remaining 200 μL was reserved for antibody labeling. The cells were stained with an allophycocyanin (APC)-conjugated anti-CD44 antibody (SAB4700180; Sigma-Aldrich, Germany) and a fluorescein isothiocyanate (FITC)-conjugated mouse anti-human CD24 antibody (560992; BD Biosciences, USA) at a concentration of 1 μL per 10⁶ cells. The samples were incubated in the dark at 4°C for a minimum of 20 minutes. Following incubation, the cells were centrifuged to remove unbound antibodies, and the pellet was washed with 500 μL of PBS. This washing procedure was repeated twice before the cells were finally resuspended in 1 mL of PBS. Both the control and labeled samples were sorted using a BD FACSMelody™ Cell Sorter (BD Biosciences, USA).
Cells demonstrating the CD44⁺/CD24⁻/low phenotype were isolated and subsequently cultured in Human Mammary Epithelial Cell Basal Medium (formerly Medium 171; Gibco, USA) enriched with 1% Mammary Epithelial Growth Supplement (MEGS; Gibco, USA) and a 1% antibiotic solution (Thermo Fisher Scientific, USA). These cultures were maintained at 37°C under normoxic, 5% CO₂ conditions. This specific subpopulation has been previously confirmed to possess robust cancer stem cell characteristics29. Prior to subsequent downstream assays, the purity and retention of the CD44⁺/CD24⁻/low phenotype within the expanded BCSC population were verified via analytical flow cytometry.
Evaluation of Cobalt Chloride (CoCl₂) Cytotoxicity for Hypoxia Modeling
To establish a stable hypoxia-mimetic model, a 25 mmol/L stock solution of CoCl₂ (Xilong Scientific, China) was prepared. BCSCs were seeded into 96-well plates (SPL, Korea) at a density of 4 × 10³ cells per well in 100 µL of medium and allowed 24 hours to adhere. The medium was then replaced with fresh medium containing varying concentrations of CoCl₂ (0, 25, 50, 75, 100, 150, 200, and 250 µmol/L). Cellular proliferation and viability were assessed at 24-, 48-, and 72-hour intervals using the alamarBlue assay (Sigma-Aldrich, Germany). Specifically, 10 µL of the alamarBlue reagent was added to each well (final concentration: 10 µg/mL), followed by a 1-hour incubation in the dark. Fluorescence was quantified at an emission wavelength of 590 nm using a DTX880 multimode plate reader (Beckman Coulter, USA). Wells containing only culture medium and the reagent served as blank controls. The percentage of cell proliferation was calculated by subtracting the blank fluorescence from the target readings and normalizing the values against the vehicle control. The mean and standard deviation (SD) for each concentration were derived from three independent biological replicates.
Quantification of HIF-1α and PHD3 mRNA Expression via RT-qPCR
BCSCs were seeded into 6-well plates at a density of 1 × 10⁵ cells per well in 1.5 mL of culture medium. After 24 hours, the control group received fresh medium without CoCl₂, while the experimental group was exposed to medium containing 75 µmol/L CoCl₂. Cells from both cohorts were harvested at 24, 48, and 72 hours post-treatment, and total RNA was extracted utilizing the easy-BLUE™ Total RNA Extraction Kit (iNtRon Biotechnology, Korea). RNA purity (OD260/OD280 and OD260/OD230 ratios) and concentration were measured using a NanoDrop ND-1000 spectrophotometer (Thermo Fisher Scientific, USA). All reactions were normalized to an input of 100 ng of total RNA.
Gene expression profiling was executed using the Luna® Universal One-Step RT-qPCR Kit (New England Biolabs, USA) according to the manufacturer’s guidelines. Reverse transcription was performed at 55°C for 30 minutes using the WarmStart® enzyme system. The qPCR cycling conditions included an initial denaturation at 95°C for 1 minute, followed by 40 cycles of denaturation at 95°C for 10 seconds and extension at 60°C for 30 seconds. The target genes included β-actin (forward: 5’- CTGGAACGGTGAAGGTGACA-3’; reverse: 5’-AAGGGACTTCCTGTAACAATGCA-3’)30, which served as a commonly used reference gene for CoCl–induced hypoxia model31,32, HIF-1α (forward: 5’-TAGCCGAGGAAGAACTATG AAC-3’; reverse: 5’-CTGAGGTTGGTTACTGTTGGTA-3’)33, and PHD3 (forward: 5’-GCCGGCTGG GCAAATACTA-3’; reverse: 5’-CCGGATAGCAAGCCACCAT-3’)34. RT‒qPCR amplification and data acquisition were performed using a Mastercycler® RealPlex 4 system (Eppendorf, Germany). Cycle threshold (Ct) values were analyzed using the Livak (2) method35. Melting curve analysis was performed to verify primer specificity.
Assessment of HIF-1α Target Gene Expression Under Hypoxia and Reoxygenation
BCSCs were seeded in 6-well plates (1 × 10⁵ cells/well) and allocated into three experimental arms: control, hypoxia, and reoxygenation. Following a 24-hour adherence period, the control group received CoCl₂-free medium, whereas the hypoxia and reoxygenation groups were treated with 75 µmol/L CoCl₂. At 72 hours, cells in the control and hypoxia groups were harvested. The reoxygenation group was subjected to a washout step and subsequently cultured in CoCl₂-free medium for an additional 24 hours to simulate rapid oxygen restoration. Total RNA was extracted from all groups using the easy-BLUE™ Kit. The mRNA expression levels of HIF-1α, PHD3, β-actin, and downstream HIF-1α targets, including VEGF36, EPO37, and ABCG238, were quantified using RT-qPCR as described above.
Determination of Cisplatin Resistance via IC Assays
A 2.5 mmol/L stock solution of cisplatin (Sigma-Aldrich, Germany) was formulated. BCSCs were seeded into 96-well plates at a density of 4 × 10³ cells per well and incubated for 24 hours. The cells were then stratified into control, CoCl₂-treated (hypoxia), and reoxygenation groups. The control group received standard medium, while the remaining two groups received medium containing 75 µmol/L CoCl₂. After 72 hours, the reoxygenation group was washed and cultured in standard medium for a further 24 hours. All experimental arms were then exposed to increasing concentrations of cisplatin (0, 1, 5, 25, 100, and 500 µmol/L). Following a 24-hour drug exposure period, cell viability was evaluated using the alamarBlue assay via a DTX880 multimode plate reader. The half-maximal inhibitory concentration (IC) values for cisplatin were calculated via non-linear regression analysis (log[inhibitor] vs. normalized response) using GraphPad Prism software (version 10.3.1).
Statistical Analysis
All quantitative data are presented as the mean ± standard deviation (SD) derived from a minimum of three independent biological replicates. Flow cytometry data were analyzed and visualized using FlowJo software (version 10.9.1). Differences between two independent groups were evaluated using an unpaired Student’s t-test, whereas comparisons involving three or more groups were assessed via a one-way analysis of variance (ANOVA) followed by Tukey’s post-hoc test. Statistical significance was defined as p < 0.05 (*), p < 0.01 (**), p < 0.001 (***), and p < 0.0001 (****). Results with a p-value > 0.05 were considered non-significant (ns). All statistical operations and graphical plotting were executed using GraphPad Prism (version 10.3.1).
Results
Isolation and Proliferation of BCSCs
VNBRCA1 cells cultured in DMEM/F12 medium supplemented with 10% FBS and 1% antibiotics exhibited a characteristic epithelial morphology (Figure 1A). Flow cytometric analysis revealed that the CD44⁺/CD24⁻/low subpopulation comprised 1.573 ± 1.167% of the total cell population (Figure 1B–D). Following expansion to a yield of 2–3 × 10⁷ cells, this CD44⁺/CD24⁻/low fraction was isolated and subsequently cultured in M171 medium enriched with 1% MEGS and 1% antibiotics. Post-sorting, these cells adopted an elongated, mesenchymal-like morphology characterized by prominent cluster formation (Figure 1E). The BCSCs were further expanded in M171 medium, and their phenotypic purity was re-evaluated prior to downstream assays. Subsequent flow cytometry confirmed that 93.427 ± 2.935% of the cultured cells successfully retained the targeted CD44⁺/CD24⁻/low BCSC phenotype (Figure 1F–H).
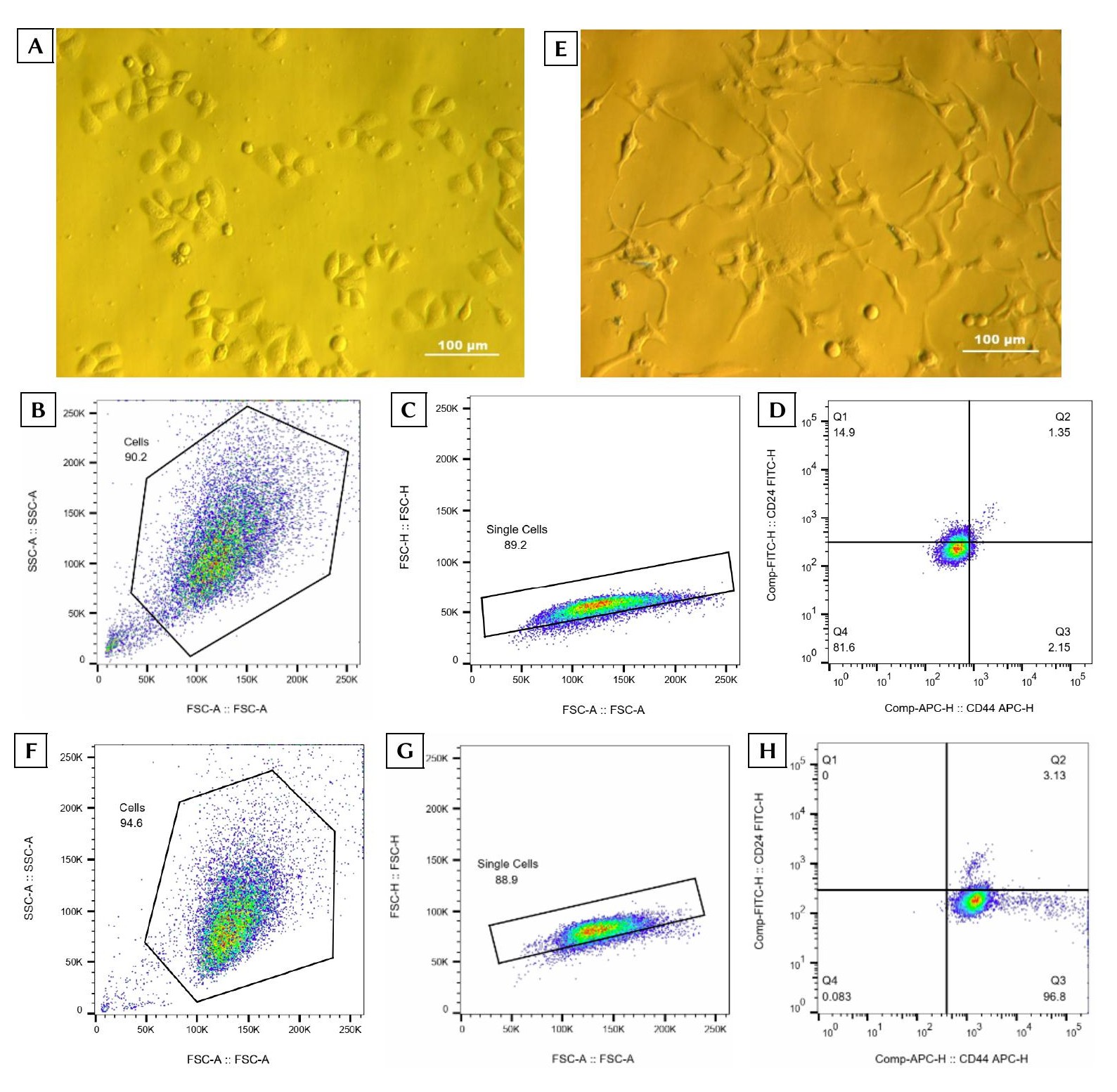
Isolation and stemness validation of breast cancer stem cells (BCSCs) derived from the VNBRCA1 cell line.(A) Representative phase-contrast image of VNBRCA1 cells cultured in DMEM/F12 medium supplemented with 10% FBS and 1% antibiotics (Zeiss Axiovert 40 C inverted microscope; Carl Zeiss AG, Germany). (B) Initial flow cytometric gating of VNBRCA1 cells identifying a homogeneous event cluster based on forward scatter area (FSC-A: 100K–<250K) and side scatter area (SSC-A: 50K–<250K) parameters. (C) Doublet exclusion and single-cell discrimination utilizing FSC-A versus FSC-H gating. (D) Quadrant analysis delineating four distinct cell subpopulations: Q1, CD24⁺/CD44⁻; Q2, CD24⁺/CD44⁺; Q3, CD24⁻/CD44⁺ (the target BCSC phenotype); and Q4, CD24⁻/CD44⁻. (E) Representative image of the sorted cancer stem cells cultured in M171 medium supplemented with MEGS and 1% antibiotics. (F) Re-gating of the expanded BCSCs as a uniform event cluster using FSC-A (100K–<250K) and SSC-A (50K–<250K). (G) Confirmation of single-cell gating via FSC-A versus FSC-H analysis. (H) Post-expansion quadrant distribution of CD44 and CD24 expression, interpreted as described in panel (D) (n = 3 independent biological replicates).
Establishment of a CoCl₂-Induced Hypoxia Model in BCSCs
To determine the optimal conditions for a prolonged hypoxia model, BCSCs were seeded into 96-well plates and stabilized for 24 hours prior to exposure to varying concentrations of CoCl₂ (0, 25, 50, 75, 100, 150, 200, and 250 µmol/L). Cellular viability was assessed over 24, 48, and 72 hours using the alamarBlue assay. Compared to the vehicle control (0 µmol/L), CoCl₂ concentrations up to 75 µmol/L did not significantly impair cell viability (p > 0.05); however, concentrations at or exceeding 100 µmol/L induced marked cytotoxicity (p < 0.05) across all time points (Figure 2A).
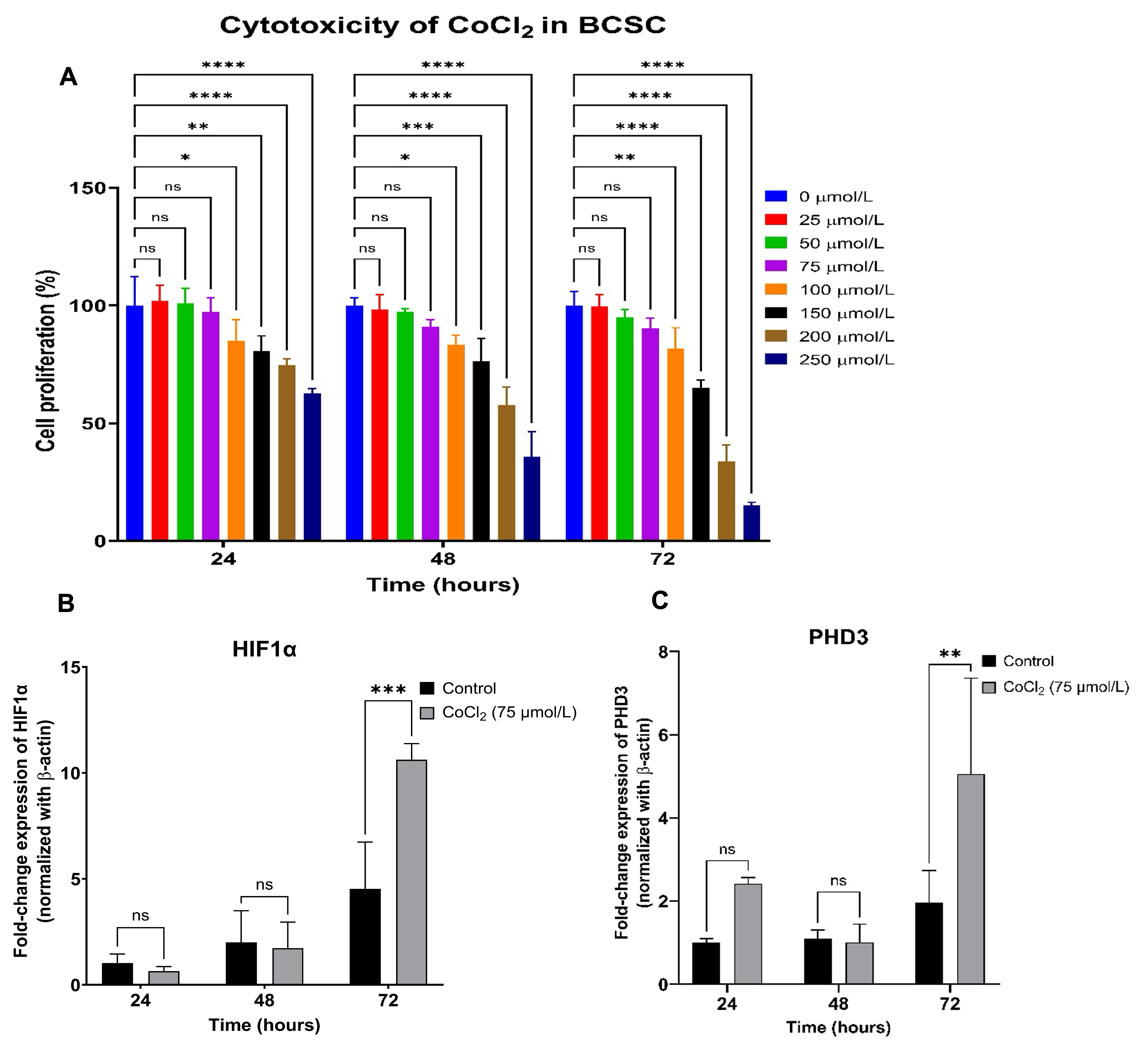
Establishment of a cobalt chloride (CoCl₂)-induced hypoxia model.(A) Cytotoxic effects of CoCl₂ on BCSCs treated with concentrations ranging from 0 to 250 µmol/L (0, 25, 50, 75, 100, 150, 200, and 250 µmol/L) evaluated at 24, 48, and 72 hours post-treatment. (B) Relative mRNA expression of
Preserving cell viability over a 72-hour exposure period was deemed critical for accurately modeling sustained hypoxia. Given that physiological tissue oxygen levels typically range from 1–5% O₂, allowing cells to remain viable while mounting adaptive hypoxic responses39, concentrations between 25 and 75 µmol/L were carefully evaluated. Ultimately, 75 µmol/L CoCl₂ was identified as the optimal concentration to maximize hypoxia-mimetic signaling while maintaining adequate cell viability.
Subsequent RT-qPCR analysis of BCSCs cultured in 6-well plates and exposed to 75 µmol/L CoCl₂ demonstrated significant upregulation of both HIF-1α and PHD3 at 72 hours compared to time-matched controls (HIF-1α: 10.62 ± 0.76 vs. 4.53 ± 2.20; PHD3: 5.05 ± 2.30 vs. 1.97 ± 0.76; p < 0.05). No statistically significant differences in the expression of these genes were observed at the 24- or 48-hour time points (Figure 2B, C). These findings confirm that a 72-hour treatment with 75 µmol/L CoCl₂ successfully establishes stable, hypoxia-mimetic conditions in BCSCs.
Expression Dynamics of Hypoxia-Responsive Genes Under Sustained Hypoxia and Reoxygenation
BCSCs were stratified into three experimental cohorts: a normoxic control, a CoCl₂-treated group (prolonged hypoxia), and a reoxygenation group. The expression levels of HIF-1α and PHD3, alongside established downstream targets regulated via hypoxia response elements (ABCG2, VEGF, and EPO), were quantified across all groups. RT-qPCR revealed a significant upregulation of HIF-1α and PHD3 in the CoCl₂-treated cohort compared to the normoxic control (p < 0.05) , followed by a pronounced downward trend in the reoxygenation group (Figure 3A, B).
Similarly, VEGF expression mirrored the patterns of HIF-1α and PHD3, showing a distinct increase under hypoxic conditions and a subsequent decline upon reoxygenation (Figure 3C). Conversely, ABCG2 and EPO exhibited a progressive downregulation from the control to the CoCl₂-treated group, reaching their lowest expression levels following reoxygenation (Figure 3D, E). This unexpected suppression of ABCG2 —a gene critically implicated in cancer cell chemoresistance—prompted further investigation into the functional impact of these culture conditions on BCSC cisplatin sensitivity.
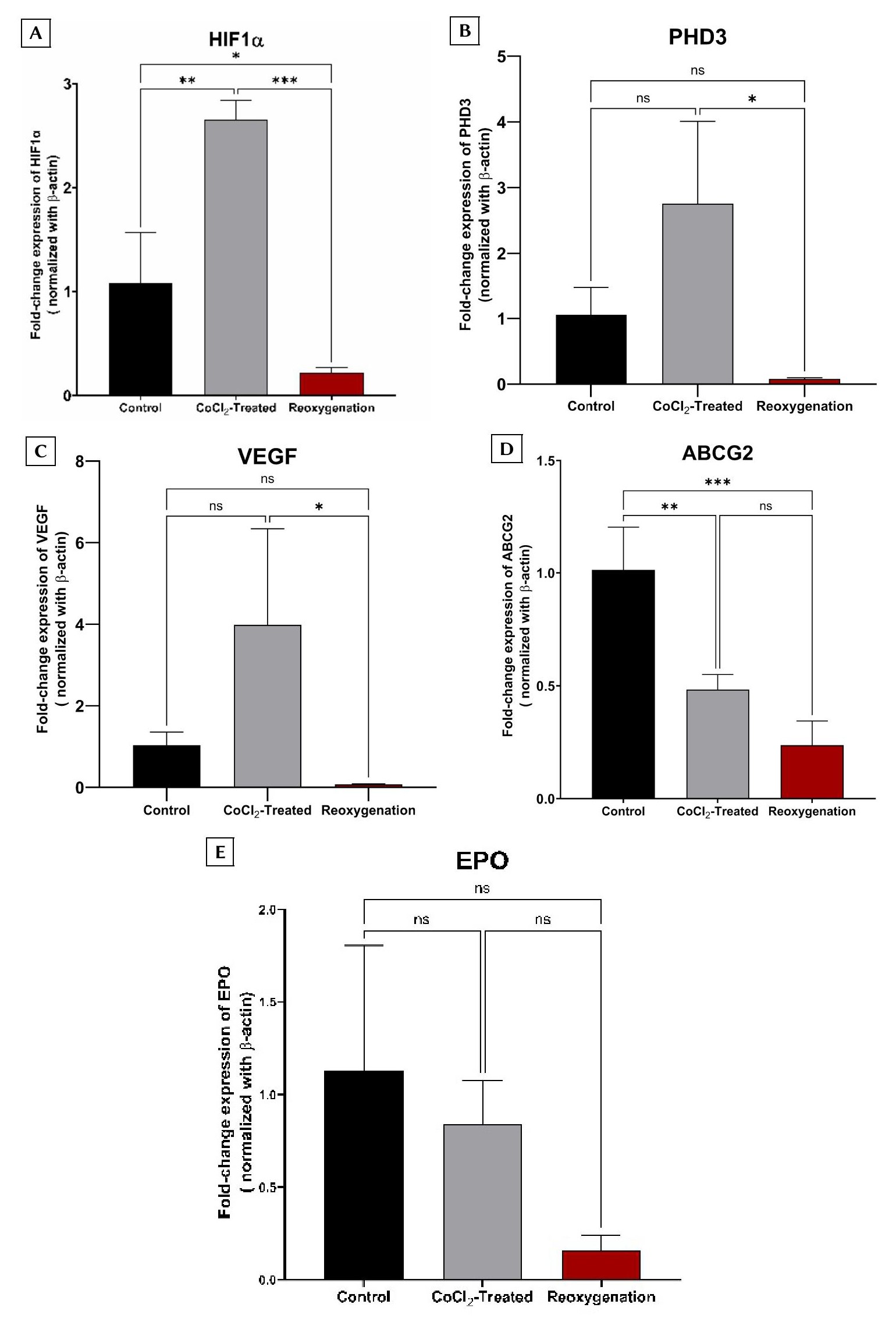
Fold-change expression of target genes across the three experimental groups. The experimental cohorts included: a control group (non-CoCl₂-treated); a CoCl₂-treated group (cells exposed to 75 µmol/L CoCl₂ for 24 h); and a reoxygenation group (cells treated with 75 µmol/L CoCl₂ for 24 h, followed by culture in CoCl₂-free medium for an additional 24 h). Relative mRNA fold-change expression is shown for: (A)
Evaluation of Cisplatin Resistance Across Culture Conditions
To assess chemoresistance functionally, BCSCs were seeded in 96-well plates, subjected to the three aforementioned culture conditions, and subsequently treated with increasing doses of cisplatin (0 to 500 µmol/L). Following 24 hours of drug exposure, relative cell survival was quantified. The calculated cisplatin IC values for the control, CoCl₂-treated, and reoxygenation groups were 122.2 ± 35.59 µmol/L, 99.44 ± 36.79 µmol/L, and 66.45 ± 26.38 µmol/L, respectively (Figure 4). Despite the observed trend toward increased cisplatin sensitivity in the reoxygenation group, one-way ANOVA indicated that these differences did not reach statistical significance among the three cohorts.
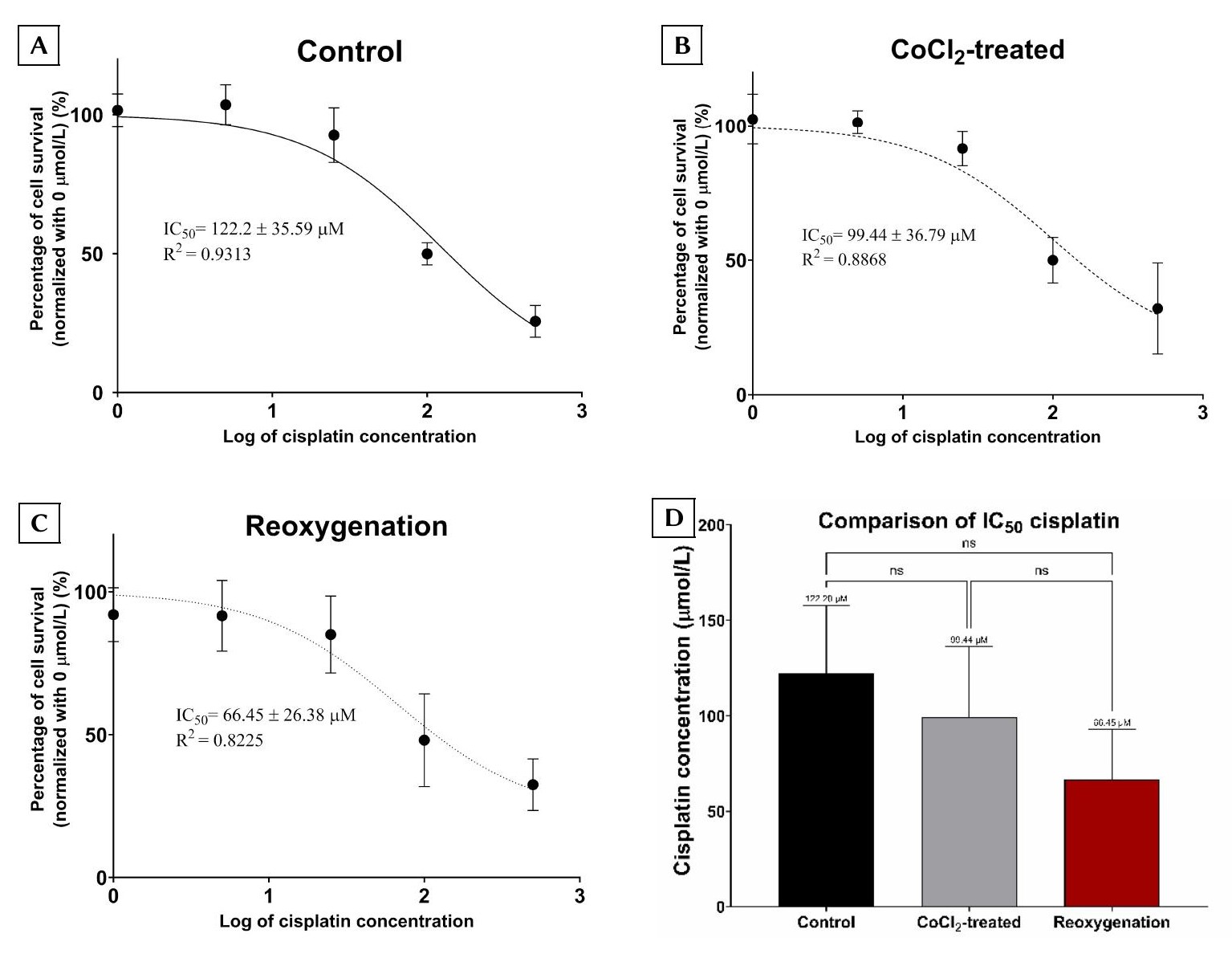
Evaluation of cisplatin resistance in BCSCs across the three experimental groups. The IC50 values for cisplatin were determined in the control (non-CoCl₂-treated), CoCl₂-treated (cells exposed to 75 µmol/L CoCl₂ for 24 h), and reoxygenation groups (cells treated with 75 µmol/L CoCl₂ for 24 h followed by culture in CoCl₂-free medium for an additional 24 h). (A) Control group; (B) CoCl₂-treated group; and (C) Reoxygenation group. (D) Comparison of the calculated IC50 values among the three experimental cohorts. Data are presented as the mean ± SD derived from at least three independent biological replicates (n = 3). Statistical significance is denoted as follows: ns, not significant (
Discussion
Cancer cells exhibiting a CD44⁺/CD24⁻/low phenotype isolated from breast tumors possess well-documented stem cell-like properties40. Similarly, side-population cells with this specific phenotype derived from the VNBRCA1 breast cancer cell line have been reported to display defining cancer stem cell (CSC) features, including tumor initiation capacity, drug resistance, and metastatic potential29. Consistent with these established findings, the present study successfully isolated and maintained CD44⁺/CD24⁻/low cells to serve as a targeted experimental model for breast cancer stem cells (BCSCs).
Cobalt chloride (CoCl₂) is widely utilized as a chemical hypoxia-mimetic agent41. This compound upregulates HIF-1α via both transcriptional and translational mechanisms42. Conversely, HIF-1α enhances PHD3 expression during sustained hypoxia by binding to the hypoxia-response element (HRE) within the PHD3 promoter43. Furthermore, previous literature demonstrates that the optimal CoCl₂ concentration for inducing hypoxia is highly dependent on the specific cancer cell line utilized7,44. In the current study, the maximal induction of both HIF-1α and PHD3 was observed following a 72-hour treatment with 75 µmol/L CoCl₂. Consequently, these optimized parameters were employed to establish a robust prolonged hypoxia model. However, it is imperative to note that CoCl₂ may exert off-target effects independent of canonical hypoxic signaling pathways. Cobalt ions are known to induce oxidative stress45 , disrupt mitochondrial function46 , and perturb other metal-sensitive cellular mechanisms47,48,49 , all of which could potentially confound interpretations specific to true hypoxia. Accordingly, findings derived from CoCl₂-based models should be interpreted with caution and warrant future validation using physiological hypoxia systems, such as specialized hypoxia chambers.
Reoxygenation entails the resupply of oxygen to cells or tissues following a period of oxygen deprivation50. While this phenomenon is frequently associated with ischemia-reperfusion cellular injury51, reoxygenation within the context of cancer research has also been shown to exert profound effects on cancer cell biology and radiotherapy outcomes52,53. Its specific role in modulating the chemoresistance of hypoxic cancer cells, however, remains incompletely elucidated. Nevertheless, some previous studies indicate that restoring oxygen to hypoxic, drug-resistant tumor cells can potentially reverse their resistance profiles, thereby re-sensitizing them to anticancer therapeutics54,55. To evaluate the impact of reoxygenation on CSC drug resistance in our model, BCSCs subjected to prolonged CoCl₂ treatment were subsequently cultured in CoCl₂-free medium to simulate a rapid reoxygenation event.
Gene expression profiling revealed that HIF-1α, PHD3, and VEGF exhibited parallel expression trajectories: elevated levels under hypoxic conditions followed by a sharp decline upon reoxygenation. This trend likely reflects the rapid degradation and downregulation of HIF-1α during reoxygenation, which subsequently curtails the transcription of its downstream targets, VEGF and PHD3. In contrast, the expression of the erythropoiesis-associated gene EPO remained relatively stable across both the CoCl₂-treated and reoxygenation groups. This stability may be attributed to the fact that EPO is predominantly regulated by HIF-2α56,57, rather than HIF-1α.
Notably, the expression of the drug efflux transporter ABCG2 decreased significantly from the normoxic control group to the CoCl₂-treated group. This hypoxia-induced downregulation contrasts with the established role of ABCG2 in mediating chemoresistance in breast cancer cells58,59,60. Consistent with our findings, a study focusing on pancreatic cancer also reported a significant reduction in ABCG2 expression under both acute and chronic hypoxic conditions61; however, that study primarily cataloged chemoresistance-related gene variations without delving deeply into the underlying molecular mechanisms. Additional investigations utilizing the BeWo placental cell line and human fetal brain endothelial cells have similarly demonstrated ABCG2 downregulation under hypoxic stress62,63. Collectively, these observations suggest that hypoxia may uniquely and negatively regulate ABCG2 in certain sensitive cell types, particularly those exhibiting pronounced stem-like characteristics.
Upon reoxygenation, ABCG2 expression remained markedly lower than in the control group ; however, no significant difference was observed relative to the CoCl₂-treated cohort. This pattern implies that rapid reoxygenation following a prolonged hypoxic period may be insufficient to immediately reverse hypoxia-induced transcriptional alterations, and it may concurrently exert a negative impact on cell viability. Furthermore, the reduction in ABCG2 expression without a corresponding, statistically significant increase in cisplatin sensitivity underscores a highly complex regulatory landscape governing chemoresistance. While a decrease in the cisplatin IC from the control to the reoxygenation group was observed, the difference failed to reach statistical significance. A post hoc power analysis suggested that the current sample size may have been underpowered to detect modest intergroup differences. The substantial variability noted in the cisplatin dose–response curves likely reflects inherent biological heterogeneity within the BCSC subpopulation, intrinsic assay variability, and the limited number of biological replicates (n = 3). Accordingly, these IC results should be considered preliminary and require validation in larger-scale studies.
Several limitations of the present study must be acknowledged. First, the utilization of CoCl₂ as a hypoxia mimetic, rather than employing specialized systems to generate physiological hypoxia, may elicit distinct cellular responses due to differing mechanisms of action. Second, additional protein-level validation of the analyzed mRNA targets, particularly HIF-1α and PHD3, is necessary to confirm that these transcriptional changes translate proportionally to the functional protein level. A critical limitation is the omission of HIF-2α assessment. While HIF-1α acts as a principal mediator of the acute hypoxic response, HIF-2α regulates a partially distinct transcriptional program critically implicated in stemness, erythropoietic signaling, and therapy resistance. The absence of HIF-2α data, therefore, restricts a fully comprehensive interpretation of hypoxia-driven regulatory networks in BCSCs. Future studies should incorporate parallel transcript- and protein-level analyses of HIF-2α to delineate isoform-specific roles and bolster the mechanistic framework. Another methodological challenge is the inherent difficulty in maintaining the highly plastic CD44⁺/CD24⁻/low stem-like phenotype over prolonged culture periods ; nevertheless, surface marker expression was rigorously verified across each experimental replicate in this study. Finally, the exclusive reliance on a single, local cell line (VNBRCA1) limits the broader generalizability of these findings across diverse breast cancer subtypes.
Conclusion
This study successfully isolated and enriched a distinct subpopulation of breast cancer cells characterized by a CD44⁺/CD24⁻/low cancer stem-like phenotype. The application of 75 µmol/L CoCl₂ over a 72-hour period effectively established a robust, prolonged hypoxia-mimetic in vitro model. The induction of hypoxia in these breast cancer stem cells (BCSCs), followed by rapid reoxygenation via medium replacement, led to a marked downregulation of the drug efflux transporter ABCG2. However, this observed molecular alteration did not translate into a statistically significant enhancement in cisplatin sensitivity. Collectively, these findings indicate that the dynamic modulation of tumor oxygenation may influence the chemotherapeutic response profile of hypoxic cancer stem cells. Nevertheless, further comprehensive investigations exploring downstream hypoxia-responsive targets, particularly EPO, are essential to fully elucidate the complex underlying molecular mechanisms governing these cellular adaptations.
Abbreviations
ABCG2: ATP binding cassette subfamily G member 2; ANOVA: Analysis of variance; ARNT: Aryl hydrocarbon receptor nuclear translocator; BCSCs: Breast cancer stem cells; bHLH: basic helix-loop-helix; CD24: cluster of differentiation 24; CD44: cluster of differentiation 44; DMEM/F12: Dulbecco's modified Eagle’s medium: nutrient mixture F12; EMT: epithelial‒mesenchymal transition; EPO: Erythropoietin; HIF-1α: hypoxia-inducible factor 1 alpha; HRE: hypoxia response element; MEGS: Mammary Epithelial Growth Supplement; PAS: PER-ARNT-SIM; PER: Period protein; PHD3: prolyl hydroxylase domain 3; SIM: Single-minded; TME: tumor microenvironment; VEGF: Vascular endothelial growth factor.
Acknowledgments
None.
Author’s contributions
Nhan Ngo-The Tran and Khan Dinh Bui contributed equally to all aspects of this work. Nhan Ngo-The Tran was primarily responsible for experimental design and execution. Khan Dinh Bui provided critical feedback and detailed revisions for the manuscript.
Funding
This research is funded by University of Science, VNU-HCM under grant number T2023-80.
Availability of data and materials
Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Declaration of generative AI and AI-assisted technologies in the writing process
The authors declare that they have used generative AI and/or AI-assisted technologies in the writing process before submission, but only to improve the language and readability of their paper.
Competing interests
The authors declare that they have no competing interests.

