Association of P73, BMP15, and GDF9 gene polymorphisms with diminished ovarian reserve
- Department of Medical Genetics, Shahid Sadoughi University of Medical Sciences, Yazd, Iran
- Department of Medical Genetics, Shahid Beheshti University of Medical Sciences, Tehran, Iran
- Abortion Research Center, Yazd Reproductive Sicences Institue, Shahid sadoughi University of Medical Sciences, Yazd, Iran
Abstract
Introduction: Diminished ovarian reserve (DOR) is a condition in which the quantity or quality of oocytes results in impaired fertility. The prevalence of DOR in infertile women is estimated to be around 10%. Our aim was to investigate the polymorphisms rs3810682, rs10491279, and rs4648551 in association with the genes BMP15, GDF9, and p73 in patients with DOR and control samples.
Methods: Genomic DNA was isolated from 5 cc of peripheral blood from 300 participants (150 patients with DOR and 150 women without a history of DOR). After ARMS-PCR assay and sequencing of a series of samples to confirm results, data were analyzed using GraphPad Prism software.
Results: A significant difference was found between the frequencies of the different polymorphisms associated with the BMP15 gene (P value = 0.002). However, for the polymorphisms of the GDF9 and p73 genes, no significant difference was found between the frequencies of these polymorphisms in the control and patient groups.
Conclusion: Determining the frequency of gene variants in case and control groups helps in understanding the potential risk factors for the development of DOR. Understanding these factors is very important in different populations that naturally have different lifestyles, especially in a country like Iran, where a large number of pregnancies occur at older ages due to the use of contraceptives. Determining risk factors can help women decide when to become pregnant. Finally, further validation is needed to obtain more information on the use of the polymorphisms investigated in this study as an indicator of DOR in Iranian populations.
Introduction
Diminished ovarian reserve (DOR) is a condition where the quantity or quality of oocytes leads to abnormal fertility1. DOR is clinically characterized by signs such as low levels of anti-Müllerian hormone (AMH) (< 0.5 – 1.1 ng/ml), low antral follicle count (AFC) (5–7 follicles), and elevated levels of follicle-stimulating hormone (FSH) (≥ 10 mIU/ml)2, 3, 4, 5. Aging is one of the main causes of DOR; smoking, and invasive treatments such as chemotherapy and radiotherapy are other causes. Genetic conditions such as fragile X syndrome, ovarian surgery, endometriosis, and autoimmune diseases are additional examples of causes of DOR6, 7. Unfortunately, most women do not exhibit any symptoms of DOR. Only as the condition progresses over time do women notice a shortening of their menstrual cycles.
In general, there is no cure to delay or prevent DOR. Reportedly, 33% of patients with DOR are able to become pregnant with their own eggs after treatment8, 9. However, reports emphasize that early diagnosis is important as it increases the chances of conception. In the meantime, the main treatment for DOR is to preserve the ability to conceive by stimulating the ovaries and using assisted reproductive techniques such as IVF10, 11. Another treatment option for women who wish to delay pregnancy is the freezing of eggs and embryos. To treat DOR, medication is administered to stimulate the ovaries to grow eggs, which can then be frozen or used for IVF. Considering the difficulty and complexity of treating diminished ovarian reserve, it seems that studying genetic and epigenetic factors could shed more light on improving our current knowledge in this field.
GDF9 encodes a secreted ligand of the TGF-beta superfamily12, 13. GDF9 is essential for the development of surrounding somatic cells, particularly granulosa, cumulus, and theca cells14. Paracrine interactions between the developing oocyte and the surrounding follicle cells are crucial for proper follicle and oocyte development. GDF9 is essential for the entire process of follicle maturation, ovulation, and egg maturation and thus plays a central role in female fertility15. GDF9 binds to the receptors for bone morphogenetic protein (BMPR) and ALK5 via two receptors on oocytes. It promotes the secretion of inhibin A, thus enhancing the ability of the follicle to develop from an early growth stage. GDF9 promotes the growth of prenatal follicles by preventing apoptosis of granulosa cells16, 17, 18. In preovulatory follicles, GDF9 induces progesterone production by stimulating the prostaglandin EP2 receptor signaling pathway. Bone morphogenetic protein 15 (BMP-15), also part of the TGF-β family, is involved in folliculogenesis and is expressed only after the early stages of oocyte development19. The promotion of ovarian follicle growth and maturation in the early stages of gonadotropin-independent folliculogenesis, the regulation of granulosa cell sensitivity to FSH, and the prevention of granulosa cell apoptosis are important functions of BMP1520. In 2004, Hanrahan and colleagues discovered a mutation in the GDF9 gene that increases ovulation and infertility rates in a manner similar to inactivating mutations in BMP1521. In 2006, Morón et al. analyzed various SNPs in the BMP15 gene in 307 Spanish women treated with rFSH. The results showed a direct correlation between the increased number of follicles and various SNPs in the BMP15 gene in response to rFSH22. In 2016, Mehdizadeh et al. investigated the association of rs3810682 of the BMP15 gene with polycystic ovary syndrome (PCOS) in the Iranian population. The results showed that the genotype was heterozygous (CG) in 28% of women with PCOS and homozygous (GG) in 2.8%. In a 2017 paper by Peluso et al., no association was found between the rs3810682 SNP of BMP15 and ovarian stimulation in Brazilian infertile women23.
In 2019, Santos et al. investigated variants of the GDF9 and BMP15 genes in women with primary ovarian insufficiency (POI) and those with high FSH levels compared to the control group. The results showed that the frequency of CT and TT genotypes in BMP15:c.852C> T (rs17003221) was higher in the POI group than in the control group, whereas the analysis of GDF9 variants showed no significant difference in genotype distribution24. In 2021, Meireles et al. conducted a study to investigate the correlation between factors associated with follicular growth and reduced ovarian response in ovaries overstimulated for IVF, such as LHR, GDF9, FSHR, AMHR2, and BMP15 polymorphisms. The results suggest that the polymorphism in the GDF9 gene may be a double-edged sword that can have both a positive (C447T) and a negative effect (398-39 C> G) on ovarian reserve25.
P73 is involved in cell cycle control and promotes apoptosis. Mouse models have shown that the p73 gene is involved in female infertility26. In 2008, Richard Tomasini et al. reported that the suppression of p73 expression in mice can lead to infertility27. Their study showed that female mice carrying deletions of exons 2 and 3 of the p73 gene had normal fertility but ovaries with reduced growth capacity and follicle size. In a 2011 study by Feng et al. on women undergoing IVF, an association was found between a SNP (rs4648551, A> G) in the p73 gene and infertility. The results showed that infertile women over the age of 35 had the “G” allele28. In 2015, a study by Vagnini et al. in 605 Brazilian women showed that a polymorphism (rs4648551, A > G) in the p73 gene could be used as a potential marker for DOR29. In the present study, our aim was to investigate the polymorphisms rs3810682, rs10491279, and rs4648551 in relation to the genes BMP15, GDF9, and p73 in patients with DOR.
Methods
After explaining the general steps of the experiment to the participants (with ethics approval code: IR.SSU.MEDICINE.REC.1398.186), 5 cc of peripheral blood was collected from 300 participants (150 patients with DOR and 150 women without a history of DOR) referred to the Yazd Reproductive Sciences Research Institute between September 15, 2020, and April 11, 2021, and placed in falcons containing 1.8 mg/ml K EDTA. Inclusion criteria included age under 35 years, normal karyotype, AMH < 2 µg/L, and no history of chemotherapy. Exclusion criteria were age over 35 years, abnormal karyotype (e.g., fragile X syndrome), and AMH > 2 µg/L. The samples were stored at -20 degrees for DNA extraction.
Genomic DNA extraction from whole peripheral blood
DNA isolation from peripheral blood was performed using the SimEX kit (Cat.: SBL15-2136) according to the manufacturer’s procedure. The concentration and purification of the DNA were tested with a spectrophotometer (Thermo Scientific, Wilmington, USA) and validated by 1% agarose gel electrophoresis.
ARMS-PCR
The ARMS-PCR assay was used to specifically amplify the sequences of interest in two tubes. A 100 ng DNA template and Amplicon Master Mix Kit as well as specific primers (Table 1) were used for amplification. The primers contained mismatches to maximize discrimination between wild-type and mutant alleles. The PCR settings were: primary denaturation at 95 °C for 1 minute, 40 cycles of denaturation at 95 °C for 30 seconds, annealing at 60 °C for 45 seconds, and extension at 72 °C for 40 seconds. The last extension was carried out at 72 °C for 10 minutes. The products were loaded onto a 2% agarose gel for 50 minutes at 110 V with a 100 bp ladder and imaged under ultraviolet light after staining with SYBR Safe DNA Gel Stain (Cat. 881603PR).
Statistical analysis
Following the ARMS-PCR assay and sequencing of a number of samples to confirm the results, statistical analysis was completed using the one-way ANOVA test followed by Dunnett’s multiple comparison test. P values less than 0.05 were considered statistically significant. Statistical analysis was performed using GraphPad Prism 6 software.
Sequence of primers used in the study
|
Gene |
Primers Sequence (5’-3’) |
Product (bp) |
|
|
F (Ex): AGCCATACTCAGTAGTTTCC |
Ex: 661 In: 396 |
|
F (In): TAGGGGCAAGTAGGATCAGG | ||
|
R (N): AGGAGGACCATCTTGAAAGG | ||
|
R (M): AGGAGGACCATCTTGAAAGC | ||
|
|
F (M): AGTCCTGCTAGAAGACTTTGGT |
Ex: 368 In: 236 |
|
F (N): AGTCCTGCTAGAAGACTTTGGC | ||
|
R (In): TTGACTTGACTGCCTGTTGTG | ||
|
R (Ex): CAGTAAGGTTGCTGGGAATT | ||
|
|
F (N): TCCGTCAGGGCTGAGGATCG |
Ex: 200 In: 129 |
|
F (M): TCCGTCAGGGCTGAGGATCA | ||
|
R (In): GGAGACAGATGTGTCTGCTGGC | ||
|
R (Ex): GCCAGTGCCAGGCCCAGCGC |

ARMS-PCR gel electrophoresis of
Abbreviations: R-N: Normalreverse; R-M: Mutated reverse

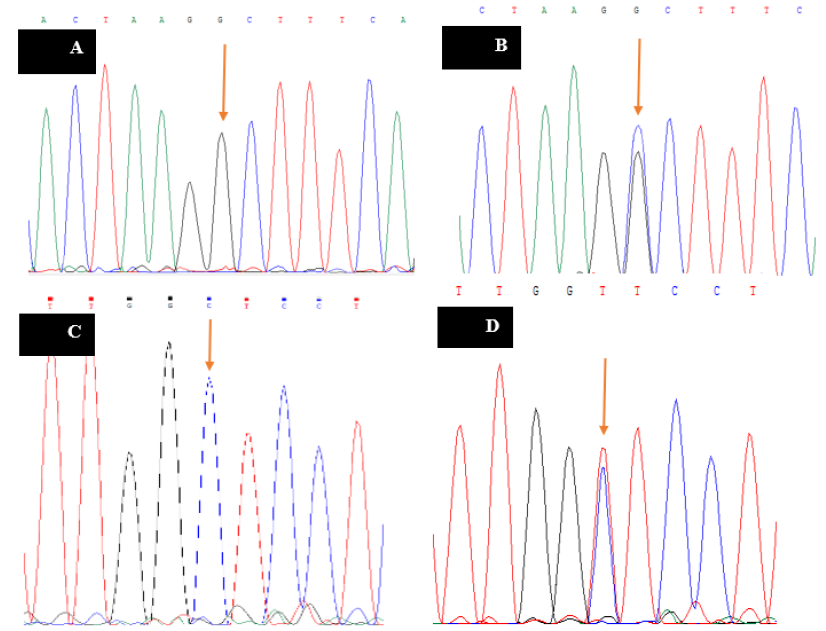
Sequencing results of a homozygous and heterozygous sample for
Results
After sampling, DNA extraction, and preparation of the PCR reaction, genotyping of samples for the BMP15, GDF9, and p73 genes was performed by ARMS-PCR in two tubes, and several samples were sequenced to confirm the results.
BMP15 Gene Genotyping
The ARMS-PCR reaction was performed for all samples, and the results were observed and analyzed on agarose gel (2%). With respect to the rs3810682 polymorphism of BMP15, the primers were designed so that the length of the PCR product of the external control primer was 661 bp and the length of the internal primer (indicating the presence or absence of the polymorphism) was 396 bp. The results of the rs3810682 polymorphism gel electrophoresis are shown in Figure 1A. This shows that in the first lane of each sample, the external control primer (product length = 661 bp) was used with the normal primer (acting with the wild-type nucleotide "C" in the samples), and in the second lane, the external control primer was used with the mutation-specific primer (G nucleotide). Therefore, 6 wells (3 samples) belonged to samples that were normally homozygous (C nucleotide), as only the lanes with the normal primer (product length = 396 bp) were used. The next four lanes (2 samples) belonged to heterozygous samples, as both normal and mutant-specific primers resulted in a PCR product. The next four wells also belonged to mutant homozygous samples. The last two wells were negative control samples for normal and mutant-specific primers. In the next step (to confirm the gel electrophoresis results), a series of samples were sequenced, and the sequencing result of this polymorphism is also shown in Figure 2A and B.
GDF9 Gene Genotyping
For the rs10491279 polymorphism of GDF9, the primers were designed so that the PCR product length of the external control primer was 368 bp, and the length of the internal primer (indicating the presence or absence of the polymorphism) was 236 bp. The gel electrophoresis results of the rs10491279 polymorphism are shown in Figure 1B, where the external control primer (product length = 368 bp) with the normal primer (acting with the wild-type nucleotide "C" in the samples) was used in the first lane of each sample, and the external control primer with the mutation-specific primer (nucleotide G) was used in the second lane. Consequently, 6 wells (3 samples) belonged to samples that were normally homozygous (C nucleotide), as only the lanes were linked to the normal primer (product length = 236 bp). The next four lanes (2 samples) belonged to heterozygous samples, as both normal and mutant-specific primers resulted in a PCR product. The next four wells also belonged to mutant homozygous samples. The last two wells were negative control samples for normal and mutant-specific primers. In the next step (to confirm the gel electrophoresis results), a series of samples were sequenced, and the sequencing result of this polymorphism is also shown in Figure 2C and D.
p73 Gene Genotyping
For the rs4648551 polymorphism associated with the p73 gene, the primers were designed so that the PCR product length of the external control primer was 200 bp, and the length of the internal primer (indicating the presence or absence of the polymorphism) was 129 bp. The gel electrophoresis results of the rs4648551 polymorphism are shown in Figure 1C. In the first lane of each sample, the external control primer (product length: 200 bp) was used with the normal primer (acting with the wild-type nucleotide "G" in the samples), and in the second lane, the external control primer was used with the mutation-specific primer (A nucleotide). Thus, 4 wells (2 samples) belonged to samples that were normally heterozygous, as both normal and mutation-specific primers resulted in a PCR product. Similarly, the next 6 lanes (3 samples) belonged to normal homozygotes (G nucleotide), as only the lanes with the normal primer (product length: 129 bp) were used. The next four wells also belonged to the mutant homozygous specimens. The last two wells were negative control samples for normal and mutant-specific primers. In the next step (to confirm the gel electrophoresis results), a series of samples were sequenced, and the sequencing result of this polymorphism is also shown in Figure 2E and F.
Discussion
The reason for DOR could be attributed to many genetic changes and is still under investigation. According to Olsen et al., various causes of DOR could lead to a reduced follicle number or a defect in the follicle-stimulating mechanisms. Regarding the rs3810682 polymorphism in BMP15, the results of the present experiment were not consistent with the results reported in some other studies, including the study by Gonzalez et al. and the study by Sproul et al.; however, it should be noted that both studies were performed in patients with polycystic ovary syndrome30, 31. In contrast, the study by Moron et al. found a statistically significant association between the high response to ovarian stimulation and BMP15 polymorphism (rs3810682), confirming the results of the present study. Decreased BMP15 levels or activity in the ovary may lead to increased FSHR expression in the granulosa cells, which could result in ovarian hyperstimulation syndrome (OHSS)32. In a study, Fonseca et al. showed that the rs3810682 in BMP15 increases the incidence of premature ovarian failure, which is due to the large number of follicles that are utilized during a woman's lifetime. A study using transgenic mice overexpressing the Bmp15 gene showed that wild-type and transgenic mice produced the same number of primary follicles, but immature transgenic mice had more atretic follicles than wild-type mice33. In general, in mice with transgenic Bmp15 expression, follicle growth was greater, atresia increased, and FSHR mRNA levels decreased34. This indicates that BMP15 increases follicle growth but not follicle maturation, which ultimately has a negative effect on ovarian reserve35.
The study by Hanevik et al. found a positive correlation between rs3810682 and OHSS36. In the present study, an inverse relationship was observed between AMH levels and the rs3810682 polymorphism; AMH levels were lower in homozygous individuals than in normal individuals. This finding contradicted the results of the study by Peluso et al. In the results of the study by Peluso et al., it was reported that AMH levels were higher in patients with the rs3810682 polymorphism than in normal individuals23. In general, polymorphisms of the BMP15 gene, especially those occurring in the promoter region, can have a significant impact on gene expression and gene function. In our study, only one of the best-known BMP15-related polymorphisms was investigated, and since BMP15 also contains other important polymorphisms, we cannot assess the effects of the entire BMP15 on infertility based on the information obtained in this study.
In this work, we investigated the rs3810682 polymorphism of BMP15, the rs10491279 polymorphism of GDF9, and the rs4648551 polymorphism of P73 in women with DOR and normal women. Individuals with elevated FSH levels and decreased AMH levels are potential candidates for DOR. The results of the DOR group were compared with the results of the control group to represent potential risk factors for DOR in the study population and to identify women at higher risk. Galloway et al. conducted a study in sheep with mutations in BMP15. In this study, a significant increase in ovulation and the birth rate of twins and triplets was observed in heterozygous models. In addition, there was ovarian failure in homozygous sheep, which could be due to incomplete follicular growth. In data from previous studies, Dixit et al. have shown that the three variants c.-9C> G, c.308A> G, and c.852C> T of BMP15 are associated with DOR37. In the case of the c.-9C> G polymorphism, Leidig et al. found no correlation between this polymorphism and DOR38. Ma et al. also found that the frequency of the C allele varied in the case group, and Dixit et al. also confirmed the association between this polymorphism and ovarian failure39. In the current study, analysis of BMP15 genotypes and alleles showed that CT and TT genotypes for BMP15: c.852 C> T were higher in the patient groups than in the control groups.
In this study, no significant difference in the frequency of the rs10491279 polymorphism was found between patients and the control group with respect to GDF9. Furthermore, no association between the polymorphism studied and DOR was found in previous studies. In 2011, Chand et al. conducted a study on 3616 individuals with normal menopause to investigate the association between 23 single nucleotide changes in five genes—AMH, AMHR2, BMP15, FOXL2, and GDF9—and menopause and evaluated rs3897937 in BMP15, the rs3810682 polymorphism being similar to our study40. In addition, they calculated six different polymorphisms related to GDF9, including rs10491279, rs30177, rs803224, rs11748063, and rs4705974, with the rs10491279 polymorphism being similar to the present study. This research team's analysis also showed no significant association between GDF9 changes and age at menopause, but rs6521896 in BMP15 was associated with late menopause.
Members of the p53 family are involved in many vital processes that contribute to the regulation of the cell cycle and apoptosis in response to DNA damage41. In addition, the p53 family has been defined as a regulatory factor for vital processes related to human reproduction42. Interpretations have shown that p73 plays an important role in maintaining follicle pool size and ovulation rate, as well as at checkpoints, and influences egg quality. Recent experiments have shown that numerous polymorphisms in this gene may play an essential role in human reproduction. In the current study, the association between the rs4648551 polymorphism and ovarian reserve in Iranian women with DOR was investigated. Since the p73 gene is related to fertility, small changes in its function could lead to individual changes in ovarian reserve and could be potential targets for reproductive diseases. However, little is known about how this polymorphism acts in female fertility. Our results showed no significant association between DOR and different genotypes of the rs4648551 polymorphism related to the p73 gene. A study conducted by Feng et al. investigated the association between this polymorphism and infertile patients aged 35 years, and the results indicated a significant association between infertility and polymorphism. Tomasini et al. had previously demonstrated a link between this gene and the number of follicles in mice. In contrast to the two studies mentioned, the present study shows that the rs4648551 polymorphism is not significantly associated with the DOR pattern.
Comprehensive genomic sequencing and GWAS studies can reveal several variations associated with DOR. Understanding these factors is essential in different populations that naturally have different lifestyles, and especially in a country like Iran where a large number of pregnancies occur at older ages due to contraceptive use. Determining risk factors can help women decide whether to become pregnant. Finally, further validation is needed to obtain more information on the use of the polymorphisms investigated in this study as an indicator of DOR in Iranian populations.
Conclusion
One of the limitations of this work is the small number of samples, which led to difficulties in the selection of cases and controls. Another limitation was the reason and method for selecting the polymorphisms of each gene, which must be such that both an innovative aspect is present and sufficient information about it was available in previous studies. The difference between the results of the current survey and the results of the different populations indicates the different ethnic composition of the Iranian population, especially the population in the Yazd urban area. There are large inter-ethnic and indigenous differences in Iran. Therefore, caution should be exercised when using international models to assign risk factors. One of the limitations of the present experiment is the number of participants, which may be increased regardless of the larger population in other demographic studies, and more informative studies may confirm the results reported in the present study with greater certainty. It is possible that other changes in the genes examined are associated with DOR but were not examined in the present study.
Abbreviations
AFC: Antral Follicle Count, AMH: Anti-Müllerian Hormone, AMHR2: Anti-Müllerian Hormone Receptor Type 2, ARMS: Amplification Refractory Mutation System, BMP15: Bone Morphogenetic Protein 15, DOR: Diminished Ovarian Reserve, FSH: Follicle-Stimulating Hormone, FSHR: Follicle-Stimulating Hormone Receptor, GDF9: Growth Differentiation Factor 9, GWAS: Genome-Wide Association Studies, IVF: In Vitro Fertilization, LHR: Luteinizing Hormone Receptor, OHSS: Ovarian Hyperstimulation Syndrome, PCR: Polymerase Chain Reaction, POI: Primary Ovarian Insufficiency, P73: p73 gene, RS: RefSeq (e.g., rs3810682), SNP: Single Nucleotide Polymorphism,TGF-β: Transforming Growth Factor Beta
Acknowledgments
The authors thank all those who participated to this experiment.
Author’s contributions
S.G, A.K and E.B performing the main steps of essay, writing the manuscript and statistical tests. S.K, S.M and M.N Collecting the samples and helping to perform DNA extraction and ARMS technique. N.G. Head of team and monitoring and fixing technical errors during all steps of the study. The authors read and approved the final manuscript.
Funding
None.
Availability of data and materials
Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
Ethics approval code: IR.SSU.MEDICINE.REC.1398.186.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.

