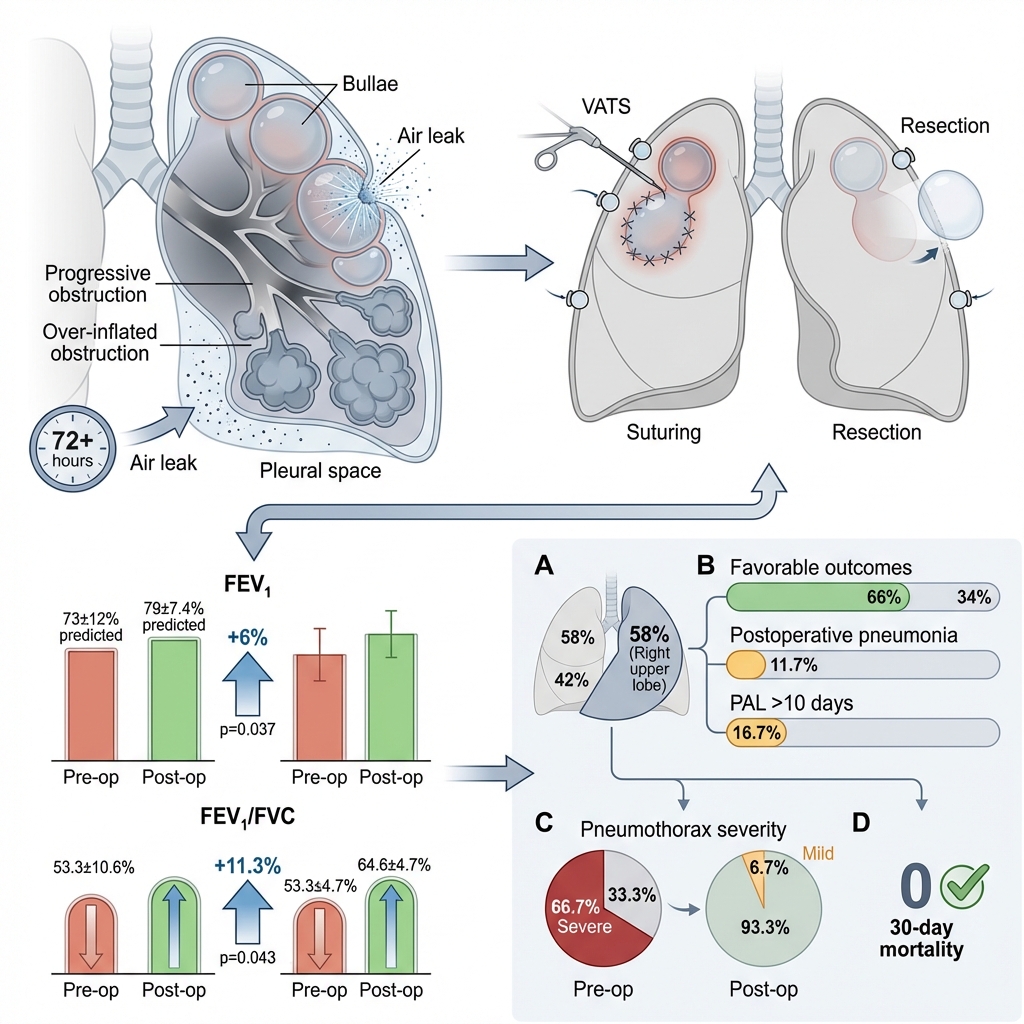
Short-term Physiological Changes After VATS for Secondary Spontaneous Pneumothorax in COPD
- Department of Thoracic Surgery, 115 People’s Hospital, Ho Chi Minh City, Viet Nam
- Southern Branch, Military Medical Academy, Viet Nam
- Department of Pulmonology, 115 People’s Hospital, Ho Chi Minh City, Viet Nam
Abstract
Objectives: Secondary spontaneous pneumothorax (SSP) is a severe complication in patients with chronic obstructive pulmonary disease (COPD) and is frequently associated with prolonged air leaks. This study aimed to describe the surgical outcomes and short-term changes in pulmonary function (PF) following video-assisted thoracoscopic surgery (VATS) for SSP in patients with COPD.
Methods: We conducted a retrospective descriptive study of 60 patients with SSP secondary to COPD who presented with a persistent air leak (PAL) lasting >72 hours following initial pleural drainage. All patients underwent VATS with bullae suturing and/or resection at 115 People’s Hospital in Ho Chi Minh City between July 2023 and December 2024. Preoperative and postoperative PF were assessed using the forced expiratory volume in one second (FEV₁) and the FEV₁/forced vital capacity (FEV₁/FVC) ratio. Surgical outcomes were categorized based on the duration of the postoperative air leak.
Results: The mean patient age was 65.4 ± 4.6 years, and the majority of the cohort consisted of smokers with localized or diffuse bullous disease identified on computed tomography. Postoperative FEV₁ increased from 73 ± 12% to 79 ± 7.4% of the predicted value (p = 0.037), and the FEV₁/FVC ratio increased from 53.3 ± 10.6% to 64.6 ± 4.7% (p = 0.043). Favorable surgical outcomes were observed in 66% of the patients. Postoperative pneumonia and prolonged air leaks occurred more frequently in patients presenting with bilateral diffuse bullae and poor nutritional status.
Conclusions: VATS with bullae suturing or resection was associated with the resolution of PAL and modest short-term improvements in PF. These changes likely reflect lung re-expansion following air leak closure rather than a true physiological improvement in the underlying COPD-related airflow limitation.
Introduction
Chronic obstructive pulmonary disease (COPD) is a progressive respiratory disorder associated with increasing morbidity and mortality. Complications of the disease include spontaneous pneumothorax, heart failure, and reduced life expectancy, rendering COPD an ongoing and major challenge in clinical management 1. Persistent airway obstruction leads to alveolar gas trapping, which impairs respiratory function—most notably evidenced by a decrease in both the forced expiratory volume in one second (FEV₁) and the FEV₁/forced vital capacity (FEV₁/FVC) ratio. Progressive air trapping results in the overdistension and thinning of alveolar walls, which may eventually rupture into the pleural cavity, precipitating a pneumothorax. Additionally, small subpleural bullae may rupture into the pleural space; this can occur bilaterally, depending on the extent of the underlying parenchymal disease 1,2.
In Vietnam, the initial management of this condition typically involves the placement of a chest tube to restore negative pleural pressure 1. When closed drainage fails to achieve lung re-expansion, or when an air leak persists beyond 72 hours, surgical resection of the offending bullae is indicated 3,4. The standard surgical approach involves the resection and suturing of bullae when a definitive bronchopleural fistula is identified, or the empirical suturing of suspected apical bullae when no obvious leak is visualized intraoperatively 3.
However, the surgical outcomes of secondary spontaneous pneumothorax (SSP) complicated by COPD have not been extensively documented in medical centers across Ho Chi Minh City, particularly concerning short-term postoperative pulmonary function (PF) 1. Because spirometric measurements obtained in the presence of a persistent air leak may underestimate true pulmonary function, postoperative changes must be interpreted with caution. Accordingly, this study aimed to assess early postoperative changes in PF (specifically FEV₁ and FEV₁/FVC) at hospital discharge following bullae suturing in patients with COPD-related SSP, as well as to describe perioperative clinical outcomes, including the incidence of contralateral bullae rupture during the course of treatment.
Methods
Study Population
This retrospective study included patients with SSP secondary to COPD who experienced a persistent air leak (PAL) lasting more than 72 hours following initial chest tube drainage. Eligible patients were treated at 115 People’s Hospital in Ho Chi Minh City between July 2023 and December 2024. Initially, 70 patients were screened for eligibility. Ten patients were excluded due to incomplete PF data (n = 5) or traumatic pneumothorax (n = 5). Ultimately, 60 patients met the inclusion criteria and were included in the final analysis. Due to the retrospective nature of the study, no formal a priori sample size calculation was performed ; however, to maximize data completeness and reliability, all eligible cases within the study period were included. The study was reported in accordance with established guidelines for retrospective observational studies.
The exclusion criteria were:
-
Primary spontaneous pneumothorax not associated with COPD.
-
Traumatic pneumothorax.
Surgical Procedure
All patients underwent general anesthesia utilizing a double-lumen endotracheal tube to facilitate single-lung ventilation during surgery. VATS was performed via a three-port approach, comprising one camera port and two working ports. Bullae larger than 1 cm on the lung surface were resected using electrocautery and subsequently closed with an Ethicon endoscopic stapler (4.1 mm cartridge) or manually sutured, contingent upon intraoperative findings. Smaller bullae (<1 cm) were compressed and directly sutured to the lung parenchyma. Pleurodesis was not routinely performed, as the surgical strategy was individualized based on intraoperative findings and concerns regarding the development of extensive pleural adhesions in advanced COPD ; however, it is noted that current international guidelines generally recommend pleurodesis to reduce recurrence rates. The primary focus of the surgical strategy was the identification and repair of air-leaking bullae while preserving the pleural space. The avoidance of routine pleurodesis aimed to minimize extensive pleural adhesions, which could complicate future thoracic interventions in patients with advanced COPD. Intraoperative air leak testing was conducted by filling the pleural cavity with sterile water. Postoperative monitoring entailed chest tube drainage and chest X-ray evaluations at 24 to 48 hours post-surgery to assess lung re-expansion and the cessation of air leaks.
Study Design and Variables
This study employed a retrospective descriptive case-series design, encompassing all patients with SSP and a documented history of COPD who required chest tube placement upon admission. PF testing was performed following initial pleural drainage and partial clinical stabilization to yield an approximate functional assessment prior to surgery. Because a PAL may have still been present at the time of testing, preoperative spirometric values were likely underestimated. This underestimation is presumably attributable to airflow loss through both the bronchopleural fistula and the chest drainage system. Consequently, preoperative spirometry in this study serves as a pragmatic clinical assessment rather than a definitive baseline measurement of COPD severity. In patients whose PAL persisted beyond 72 hours, chest computed tomography (CT) was utilized to localize pulmonary bullae, which were subsequently managed surgically via resection or suturing. Upon hospital discharge, postoperative PF was reassessed, and outcomes were documented. The following variables were collected and analyzed:
-
Demographic characteristics.
-
Type of surgical technique (resection or suturing).
-
Extent of pneumothorax.
-
Body mass index (BMI), with malnutrition defined as a BMI < 18.5 kg/m².
-
Duration of surgery and length of hospital stay.
-
PF test results (FEV₁, FVC).
Formal COPD severity stratification, whether via GOLD stages or CT-based emphysema scoring, was not routinely available and thus was excluded from the analysis. Spirometry was conducted by trained technicians utilizing a single, routinely calibrated hospital spirometer in accordance with American Thoracic Society/European Respiratory Society standards. Postoperative care adhered to a standardized clinical pathway that included respiratory physiotherapy. All eligible cases were incorporated into the analysis, with no missing data identified for the variables under review. Categorical variables were reported as frequencies and percentages, while continuous variables were expressed as the mean ± standard deviation (SD). The distribution of continuous variables was evaluated using the Shapiro–Wilk test. Differences in categorical variables (e.g., clinical features and COPD status) were analyzed via the chi-square test (χ²) or Fisher’s exact test when expected cell counts were < 5; meanwhile, pre- and postoperative PF values were compared using the paired Student’s t-test for normally distributed data or the Wilcoxon signed-rank test for non-normally distributed data. The association between the surgical technique utilized and the operative time was assessed using a one-way analysis of variance (ANOVA) , with post-hoc comparisons planned only if the overall ANOVA yielded statistical significance.
The primary outcome was predefined as the short-term change in the FEV₁/FVC ratio between the preoperative assessment and hospital discharge, which reflects immediate postoperative physiological status rather than definitive postoperative lung function. Because spirometry was conducted during the early postoperative period, results may have been confounded by factors such as postoperative pain, restricted chest wall expansion, and the administration of analgesic medications. Changes in FEV₁ were evaluated as secondary outcomes. The minimal clinically important difference (MCID) for FEV₁ in patients with COPD has been documented to range between 100 and 140 mL, or approximately 4–7% of predicted values. Given the acute clinical setting, it was anticipated that any changes in FEV₁ would be modest and might not meet the established MCID thresholds for COPD ; therefore, observed alterations should be interpreted as short-term physiological variations rather than definitive clinical improvements. All statistical analyses were performed using SPSS software version 23.0 (IBM Corp., Armonk, NY, USA). A p-value of < 0.05 was considered statistically significant.
Outcome Definitions
Postoperative outcomes were evaluated exclusively during the index hospitalization and at the time of hospital discharge. In addition to descriptive outcome categories, standardized postoperative endpoints were utilized. PAL was defined as an air leak persisting for more than 5 postoperative days, aligning with commonly accepted thoracic surgery criteria. Postoperative complications were graded using the Clavien–Dindo classification system; pneumonia requiring intravenous antibiotics was categorized as a grade II complication, and no grade III–V complications were observed. Thirty-day postoperative mortality was monitored as a primary safety endpoint. For descriptive purposes, postoperative outcomes were stratified as follows:
-
Good outcome: Resolution of the air leak within ≤ 5 postoperative days, absence of major complications, and discharge within approximately 3 to 5 postoperative days.
-
Moderate outcome: PAL lasting > 5 days but ≤ 10 days, and/or the presence of Clavien–Dindo grade II complications.
-
Poor outcome: PAL extending > 10 days, persistent pneumothorax identified on imaging, or infectious complications necessitating prolonged hospitalization.
The extent of the pneumothorax was categorized as follows:
-
Small: Air volume < 1/3 of the hemithorax.
-
Moderate: Air volume ≈ 1/3 of the hemithorax.
-
Large: Air volume > 1/3 of the hemithorax.
Sample Size and Reporting Guidelines
The sample size was predicated on the total number of patients meeting the inclusion criteria at 115 People’s Hospital during the study period (July 2023–December 2024). Owing to the retrospective design of the study, no formal a priori sample size calculation was conducted. However, to optimize data completeness and ensure reliability, all eligible cases identified within the specified timeframe were included. This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for retrospective observational studies.
Results
Patient Characteristics
A total of 60 patients diagnosed with SSP secondary to COPD were included in the study. The cohort comprised 40 males and 20 females, yielding a male-to-female ratio of 2:1. The majority of patients (75%) were over 60 years of age, with ages ranging from 57 to 78 years (mean: 65.4 ± 4.6 years). Pulmonary function was assessed following initial pleural drainage using spirometric parameters, specifically the forced expiratory volume in one second (FEV₁) and the FEV₁/FVC ratio, to characterize airflow limitation. As summarized in Table 1, the cohort consisted predominantly of elderly males with a heavy smoking history. Patients demonstrated moderate airflow limitation, with a mean FEV₁/FVC ratio of 53.3 ± 10.6%, and had a low mean body mass index (BMI) of 18.3 ± 2.5 kg/m².
Baseline demographic and clinical characteristics of 60 patients with SSP associated with COPD.
| Variable | Male (n=40) | Female (n=20) | Total (n=60) |
|---|---|---|---|
| Mean Age (years ± SD) | 63.1 ± 5.3 | 72.5 ± 4.4 | 65.4 ± 4.6 |
| Body Mass Index (BMI ± SD) | 17.5 ± 2.3 | 20.0 ± 2.1 | 18.3 ± 2.5 |
| Smoking >30 pack-years (cases) | 38/40 | 12/20 | 50/60 |
| FEV₁/FVC (% ± SD) | 51.3 ± 16.6 | 55.3 ± 10.7 | 53.3 ± 10.6 |
| FEV₁ (% predicted ± SD) | 69.0 ± 11.3 | 76.0 ± 12.7 | 73.0 ± 12.0 |
Clinical Features and Bulla Distribution
All patients presented with dyspnea and respiratory distress upon admission. A persistent air leak (PAL) lasting more than 72 hours following chest tube drainage prompted further evaluation. Chest computed tomography (CT) identified pulmonary bullae in all cases. The distribution of pulmonary bullae by anatomical location is detailed in Table 2, demonstrating that the right upper lobe was the most frequently involved site (58%). The mean bulla size ranged from 1.5 to 2.0 cm. Localized bullous lesions were identified in 33 patients, whereas 27 patients exhibited diffuse bullous disease. Among those with diffuse disease, 10 patients presented with bilateral involvement. (An illustrative example of right-sided SSP in a patient with COPD is provided in Figure 1.)
Distribution of pulmonary bullae by lobe and lesion type in patients with SSP due to COPD.
| Lobe | Bulla Type | Number of Patients | Percentage (%) |
|---|---|---|---|
| Right upper lobe | Localized | 20 | 58% |
| Diffuse | 15 | ||
| Left upper lobe | Localized | 8 | 33% |
| Diffuse | 12 | ||
| Right lower lobe | Localized | 5 | 9% |
| Total | 60 | 100% |
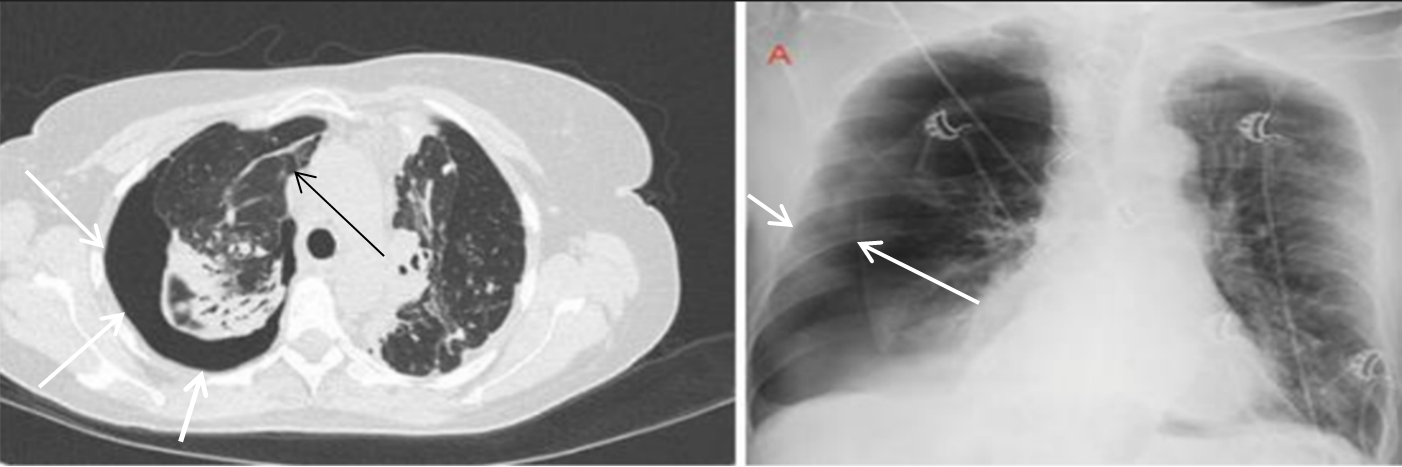
A 62-year-old man with right pneumothorax and combined pulmonary emphysema. Chest CT and X-ray images (August 2023) show right-sided SSP in a patient with COPD (white arrowheads), with a visible bullae in the right lung field (black arrowhead).
Treatment Outcomes
All 60 patients underwent VATS and/or suturing of the pulmonary bullae. As detailed in Table 3, favorable postoperative outcomes were achieved in 40 patients, whereas 10 patients each were classified as having moderate or poor outcomes based on the duration of the postoperative air leak and radiographic findings at discharge. There was no statistically significant difference in mean operative time among the various surgical techniques utilized (bullectomy resection and suture, atypical resection, and bullectomy suture only; ANOVA, p = 0.056). Because the overall ANOVA did not reach statistical significance, post-hoc comparisons were not performed. Among patients with diffuse bullous disease, 45% required a non-anatomical wedge resection or bulla suturing, whereas 55% of patients with localized lesions did not require an atypical resection.
Treatment outcomes, surgical methods, operative time, and pulmonary function changes before and after VATS in patients with SSP secondary to COPD.
| Total hospital stay, days (including preoperative drainage period): 14.4 | |||||
|---|---|---|---|---|---|
| Severity of Pneumothorax Before and After Surgery | |||||
| Severity | Preoperative n (%) | Postoperative n (%) | |||
| Mild | 2 (3.3%) | 4 (6.7%) | |||
| Moderate | 18 (30.0%) | 0 (0%) | |||
| Severe | 40 (66.7%) | 0 (0%) | |||
| Postoperative Outcomes | |||||
| Outcome | No. of patients (%) | ||||
| Good | 40 (66.6%) | ||||
| Moderate | 10 (16.7%) | ||||
| Poor | 10 (16.7%) | ||||
| Postoperative Endpoints | No. of patients (%) | ||||
| PAL >5 days | 20 (33.3%) | ||||
| PAL >10 days | 10 (16.7%) | ||||
| POP (Clavien -Dindo II) | 7 (11.7%) | ||||
| 30-day mortality | 0 (0%) | ||||
| Pulmonary Function Before and After Surgery for Air Leak Repair (n=60) | |||||
| Parameter | Preoperative (mean ± SD) | Postoperative (mean ± SD) | Mean change ± SD (95% CI) | p-value | |
| FEV₁/FVC (%) | 53.3 ± 10.6 | 64.6 ± 4.7 | +11.4 ± 9.7 (95% CI: 8.8–13.8) | 0.043 | |
| FEV₁ (% predicted) | 73.0 ± 12.0 | 79.0 ± 7.4 | +6.2 ± 5.1 (95% CI: 4.7–7.2) | 0.037 | |
| Surgical Methods and Operative Time (n=60) | |||||
| Procedure | Diffuse Lesion (n) | Localized Lesion (n) | Mean operative time (min, mean ± SD) | p-value | |
| Bullectomy (resection & suture) | 0 | 23 | 36.2 ± 4.4 | ||
| Non-anatomical wedge resection | 20 | 0 | 45.1 ± 3.7 | 0.056 | |
| Bullectomy (suture only) | 7 | 10 | 40.8 ± 9.4 | ||
Postoperative pulmonary function testing at the time of discharge revealed statistically significant improvements compared with preoperative values. The mean FEV₁/FVC ratio increased from 53.3 ± 10.6% to 64.6 ± 4.7% (p = 0.043), and the mean FEV₁ increased from 73.0 ± 12.0% to 79.0 ± 7.4% of predicted values (p = 0.037). However, given the known measurement bias associated with the presence of a PAL, these statistical differences should not be strictly interpreted as evidence of true physiological improvement. The absolute increase in FEV₁ was modest (+6.2 ± 5.1% predicted); the clinical significance of this change, relative to established minimal clinically important difference thresholds, is addressed in the Discussion section.
Postoperative complications were recorded during the index hospitalization. Pneumonia occurred in 7 of the 60 patients (11.7%) and was observed predominantly among those with bilateral diffuse bullous disease. A PAL lasting more than 10 postoperative days occurred in 10 patients (16.7%). At the time of discharge, no patients remained in the moderate or severe complication categories. No in-hospital or 30-day mortality was observed. Due to the retrospective study design and the lack of standardized long-term follow-up, the recurrence of pneumothorax, hospital readmission rates, and long-term pulmonary function outcomes could not be systematically evaluated. Furthermore, the good/moderate/poor outcome classification was utilized solely for descriptive purposes and does not represent a formally validated outcome scoring system.
Discussion
Patient Demographics and Risk Factors
International studies have demonstrated that SSP in patients with COPD is associated with elevated complication rates and predominantly affects the elderly population 5. In our cohort, gender disparities were evident, likely reflecting differences in smoking behaviors. Passive smoking and the increasing prevalence of active tobacco use among females, particularly in younger demographics, may contribute to the rising incidence of COPD in women. Notably, 60% (12/20) of the female patients in our study reported a history of smoking. Rhee et al. 2 reported that individuals who smoke possess a 4.28-fold greater risk of developing COPD compared to non-smokers. The mean body mass index (BMI) in our cohort was 18.3 ± 2.5 kg/m², indicative of a high prevalence of malnutrition. Previous research has shown that COPD complicated by malnutrition is associated with an odds ratio (OR) of 3.5 (95% CI: 2.9–4.2) for adverse outcomes, which escalates to 6.7 (95% CI: 2.3–6.2) in severely malnourished individuals 2. The underlying pathophysiological mechanism is thought to involve excessive energy expenditure by respiratory muscles and mechanical compression of the gastrointestinal tract by hyperinflated lungs, both of which contribute to anorexia and a caloric deficit.
Clinical and Paraclinical Characteristics
In patients with COPD, SSP serves as a marker of advanced disease and is associated with increased mortality. Multiple studies have established that advanced age, emergency transport to tertiary centers, and prolonged hospitalization contribute to an elevated mortality risk in this population 6. In the present study, patients underwent chest tube drainage for SSP with air leaks persisting for more than three days. Upon the resolution of acute dyspnea, spirometry was performed, revealing mean FEV₁ and FEV₁/FVC values of 73.0 ± 12.0% and 53.3 ± 10.6%, respectively, which correspond to moderate COPD severity. Literature suggests that patients experiencing recurrent pneumothorax exhibit a significantly greater decline in FEV₁ and/or FEV₁/FVC compared to those without recurrence following surgical intervention 7. Furthermore, the compromised blood oxygen reserve in COPD—even following pleural drainage—predisposes patients to critical clinical deterioration. These individuals remain at an elevated risk for aspiration events, superimposed pulmonary or pleural infections, and the exacerbation of comorbidities such as right heart failure, electrolyte disturbances, and general wasting. Previous reports indicate that mortality is higher among COPD patients with SSP compared to those with SSP unrelated to COPD 6. In clinical practice, sepsis secondary to pneumonia or empyema is a leading cause of death due to acute respiratory failure following chest drainage. It should be noted that the current study population consisted primarily of patients with moderate COPD (mean FEV₁: 73 ± 12% predicted). Individuals with severe COPD, advanced cachexia, chronic corticosteroid use, or multiple significant comorbidities may have been underrepresented, thereby limiting the generalizability of these findings and highlighting the need for further investigation in broader, more diverse cohorts.
Discussion on Treatment Approach
The primary goals in the treatment of SSP are to re-establish negative pleural pressure, promote lung re-expansion, restore respiratory function, and mitigate the risk of recurrence. In patients with COPD and a persistent air leak (PAL), chest drains often require prolonged placement compared to non-COPD patients 5. Surgical management entails the resection or suturing of visceral pleural bullae utilizing thoracoscopic staplers, clips, or non-anatomic (wedge) lung resections targeting the diseased parenchyma. The decision to pursue surgical intervention must be highly individualized, carefully weighing PF, comorbidities, and overall operative risk. Multidisciplinary consultation involving anesthesiology and respiratory medicine is paramount for optimizing perioperative planning. Minimally invasive approaches, such as VATS, are generally preferred due to their associated benefits, which include shorter operative times and reduced postoperative pain. Current international guidelines recommend considering definitive procedures like pleurodesis to lower the recurrence rate of SSP 8,9. However, these recommendations are conditional and hinge on the patient's clinical status and surgical risk profile. In patients with advanced COPD and diffuse emphysematous changes, the extensive pleural adhesions induced by pleurodesis may complicate future thoracic interventions. Consequently, the surgical strategy employed in the present study focused on the targeted treatment of air-leaking bullae while eschewing routine pleurodesis to preserve the pleural space.
It is acknowledged that VATS possesses limitations in patients with dense pleural adhesions or complex pathologies, scenarios in which an open thoracotomy provides superior exposure. Notably, recurrence rates have been reported to be comparable between open and thoracoscopic approaches 10,11. In our cohort, localized bullae—frequently situated at the apices—were effectively managed using endoscopic staplers. Facilitated by double-lumen endotracheal tubes for single-lung ventilation, the operative time for bullae management ranged from approximately 31 to 46 minutes. Conversely, 45% of the patients presented with diffuse bullous disease, which posed significant intraoperative challenges. Treatment in these instances was dictated by the surgeon's intraoperative assessment and involved either wedge resection or direct suturing of the air-leaking bullae with nonabsorbable sutures to compress the compromised region. Diffuse, pathologically distended lung parenchyma carries an elevated risk of PAL if adjacent tissue is inadvertently resected or traumatized during suturing 12,13,14. Furthermore, pulmonary parenchymal resection can compromise respiratory reserve and impair overall PF 15,16. In patients with diffuse lung involvement, additional parenchymal resection threatens to further diminish an already tenuous lung reserve and exacerbate postoperative PF decline. As a result, these patients typically endured longer operative times, required extended postoperative monitoring, and experienced less favorable clinical outcomes compared to those with localized disease. Conversely, patients with isolated bullae achieved more favorable postoperative results, which supports the feasibility and short-term procedural success of VATS bullae resection in this specific subgroup, though it does not establish definitive long-term effectiveness.
Postoperative Outcomes
The clinical trajectory of SSP in patients with preexisting COPD is inherently complex, compounded by compromised lung reserve and profound anatomical alterations. The presence of contralateral bullae or widespread emphysematous changes further complicates operative planning and adversely affects outcomes 17. In the present cohort, 66% of patients achieved favorable surgical outcomes, whereas 34% experienced moderate to poor results. Among those with poor outcomes, prolonged hospital stays were pervasive, underscoring the formidable challenge of resolving persistent air leaks. Postoperative pneumonia was frequently documented in patients exhibiting bilateral or contralateral localized pneumothoraces alongside diffuse parenchymal lesions. Of the 10 patients categorized with poor outcomes, 7 had undergone non-anatomic wedge resections, and 3 exhibited persistent pneumothoraces following bullae compression and suturing—a situation likely attributable to underlying parenchymal fragility rather than a definitive procedural failure. These refractory cases necessitated prolonged chest tube drainage (ranging from 12 to 16 days) and extended inpatient care. However, no mortalities occurred within 30 days postoperatively.
All patients received follow-up and clinical re-evaluation at the Department of Pulmonary Medicine post-discharge. Prior to discharge, all patients underwent repeat PF testing. Both FEV₁ and the FEV₁/FVC ratio demonstrated statistically significant improvements relative to preoperative baselines. Specifically, mean FEV₁ increased from 73.0 ± 12.0% to 79.0 ± 7.4% of predicted values (mean change: +6.2 ± 5.1%; 95% CI: 4.7–7.2%; p = 0.037), while the FEV₁/FVC ratio improved from 53.3 ± 10.6% to 64.6 ± 4.7% (mean change: +11.4 ± 9.7%; 95% CI: 8.8–13.8%; p = 0.043). Although statistically significant, these improvements must be interpreted cautiously given potential confounding variables, including the concurrent administration of bronchodilators, systemic corticosteroids, and antibiotics. Furthermore, the observed increase in FEV₁ falls slightly below the widely accepted minimal clinically important difference (MCID) for COPD (approximately 4–6% predicted) 1. Nonetheless, even modest augmentations in FEV₁ and the FEV₁/FVC ratio likely reflect a degree of short-term physiological stabilization in patients with severely restricted pulmonary reserve, rather than a definitive reversal of underlying disease severity. It is crucial to emphasize the observational nature of these findings, which preclude the determination of causality; the observed functional improvements cannot be conclusively attributed to the surgical intervention alone. In the absence of a matched non-surgical control group, these short-term changes in FEV₁ and FEV₁/FVC must be contextualized against the anticipated natural recovery trajectory following tube thoracostomy. Literature suggests that PF following chest tube drainage without surgical intervention in COPD-related SSP typically remains stagnant or exhibits only marginal improvement during the acute hospitalization phase. Thus, the magnitude of the functional enhancements observed herein likely indicates postoperative physiological stabilization that supersedes spontaneous recovery.
The classification schema for postoperative outcomes (good, moderate, and poor) relied on clinical and radiographic criteria and lacks formal validation, potentially introducing subjectivity into outcome adjudication. While this categorical framework was employed for descriptive clarity, standardized endpoints—including PAL > 5 days, Clavien–Dindo complication grading, and 30-day mortality—were rigorously applied to bolster objectivity and facilitate cross-study comparability. Collectively, these preliminary findings furnish valuable insights into the management of PAL in COPD-associated SSP. Onuki et al. 6 reported that the surgical management of PAL in this specific demographic carries a mortality rate of 1.9% to 4.6%.
The personalization of surgical decision-making remains a formidable obstacle. Despite comparable clinical and spirometric profiles, patient responses to surgical intervention are highly heterogeneous, heavily influenced by age, sex, comorbidities, and baseline lung reserve 18. This inherent variability often engenders clinical hesitancy regarding surgery, driven by concerns over recurrence or severe perioperative complications. In recent years, a paradigm shift toward minimally invasive thoracic surgery has been observed, correlating with enhanced outcomes and improved patient satisfaction. Furthermore, the advent of novel intraoperative adjuncts, such as biological sealants and synthetic pleural patches (e.g., poly-ε-caprolactone), has demonstrated encouraging efficacy in sealing refractory air leaks and curtailing recurrence rates 19. On a global scale, advanced therapeutic modalities—including lung volume reduction surgery and the deployment of endobronchial valves for the segmental isolation of air leaks—are actively being investigated for refractory cases 20. Lung transplantation remains a viable, albeit highly restricted, definitive solution for highly selected candidates 21. Postoperatively, comprehensive pulmonary rehabilitation, targeted respiratory physiotherapy, and structured functional exercise regimens should be prioritized as essential supportive measures to optimize lung expansion and expedite recovery 22,23.
Several limitations necessitate consideration when interpreting the findings of the present study. First, the retrospective, single-center, case-series design inherently lacks a non-surgical comparator arm, and routine pleurodesis was omitted from the surgical protocol. The absence of long-term follow-up data precludes the evaluation of recurrence risk, effectively restricting causal inferences and the formal assessment of comparative effectiveness. Consequently, the observed postoperative enhancements in PF should be viewed through the lens of expected physiological recovery post-thoracostomy, rather than being definitively ascribed to the VATS intervention. Moreover, the assessment of PF and clinical outcomes was confined to the index hospitalization, yielding a truncated observation window. Early postoperative spirometric measurements are susceptible to the confounding effects of incisional pain, restricted chest wall excursion, and opioid- or analgesic-induced respiratory suppression, all of which can artificially induce a restrictive spirometric pattern. Therefore, these findings represent transient short-term physiological fluctuations rather than enduring postoperative PF, and the limited follow-up period precluded the evaluation of recurrence, long-term functional durability, or late complications. Additionally, the limited sample size and the low incidence of specific outcome events precluded the use of multivariable regression analysis, which was intentionally avoided to prevent model overfitting; thus, residual confounding remains a distinct possibility. The study's inclusion criteria were specifically narrowed to patients with a PAL following initial chest drainage, likely selecting for a higher-risk cohort and potentially compromising the external validity and generalizability of the results. The single-center execution of this research also warrants caution before extrapolating these conclusions to disparate clinical environments. A salient methodological limitation involves the timing of the preoperative spirometry, which was conducted post-chest tube insertion but prior to definitive surgical repair. Consequently, the documented postoperative augmentation in pulmonary function may partially reflect the mechanical benefits of air leak closure and subsequent lung re-expansion, rather than a genuine amelioration of the underlying COPD-induced airflow obstruction. Future prospective investigations featuring larger sample sizes, appropriate control groups, and extended longitudinal follow-up are imperative to definitively elucidate the clinical efficacy and long-term impact of surgical management in this vulnerable patient population.
Conclusion
Secondary spontaneous pneumothorax (SSP) complicated by a persistent air leak (PAL) in patients with preexisting chronic obstructive pulmonary disease (COPD) represents a substantial clinical challenge due to the patients' compromised pulmonary and oxygen reserves. The findings of this study indicate that surgical interventions—specifically, video-assisted thoracoscopic surgery (VATS) with bullae resection and suturing—are associated with short-term postoperative changes in pulmonary function (PF) measured at hospital discharge. Postoperatively, the mean forced expiratory volume in one second (FEV₁) reached 79.0 ± 7.4% of predicted values, and the FEV₁/forced vital capacity (FVC) ratio increased to 64.6 ± 4.7% (p < 0.05).
However, because these spirometric measurements were obtained during the early postoperative period, they may have been transiently confounded by factors such as incisional pain and reduced respiratory effort. Given the brief follow-up duration and the absence of post-discharge functional assessments, these results should not be interpreted as evidence of sustained physiological improvement or definitive long-term treatment efficacy. The surgical strategy employed herein emphasized the targeted management of air-leaking bullae over routine pleurodesis, reflecting a conservative approach designed to preserve the pleural space in patients with advanced COPD.
Particular caution is warranted for patients presenting with diffuse bullous lesions and concurrent contralateral pneumothoraces—even if minimal or localized—as this cohort appears to be at a heightened risk for postoperative complications, particularly pneumonia. This risk is potentially amplified in malnourished individuals and those with a significant smoking history. Consequently, individualized perioperative decision-making and vigilant postoperative surveillance remain essential to optimizing clinical outcomes in this highly vulnerable patient population.
Abbreviations
ANOVA: Analysis of variance; BMI: Body mass index; CI: Confidence interval; COPD: Chronic obstructive pulmonary disease; CT: Computed tomography; FEV₁: Forced expiratory volume in one second; FVC: Forced vital capacity; GOLD: Global Initiative for Chronic Obstructive Lung Disease; MCID: Minimal clinically important difference; OR: Odds ratio; PAL: Persistent air leak; PF: Pulmonary function; SD: Standard deviation; SSP: Secondary spontaneous pneumothorax; STROBE: Strengthening the Reporting of Observational Studies in Epidemiology; VATS: Video-assisted thoracoscopic surgery.
Acknowledgments
The authors thank People’s Hospital 115, Ho Chi Minh City, the Department of Thoracic Surgery, the medical staff, and the patients for their support in this study.
Author’s contributions
Dong Duc Hung contributed to the study conception, surgical procedures, and manuscript drafting. Nguyen Phuc Minh Tue was responsible for data acquisition, literature review, and statistical analysis. Tran Van Song supervised the research process, provided critical revisions, and approved the final manuscript. All authors read and approved the final version of the manuscript.
Funding
None.
Availability of data and materials
Data and materials used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
This retrospective study was conducted in accordance with the principles of the Declaration of Helsinki. The study protocol was reviewed and approved by the Ethics Committee of 115 People’s Hospital, Ho Chi Minh City, Vietnam (660/BVND115-NCKH). As this was a retrospective review of medical records, the requirement for individual patient consent was waived by the Ethics Committee. All patient data were anonymized and kept confidential throughout the study.
Consent for publication
Not applicable.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work the authors used ChatGPT (OpenAI) in order to assist with English language editing and improving clarity of expression. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the published article.
Competing interests
The authors declare that they have no competing interests.

